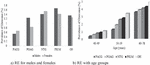Consanguinity Among Refractive Error, Intraocular Pressure and Etiology of Glaucoma in Diabetics Among Wheatish Population
Ravi Prasad Ravuri
Sriven Technologies, India
E-mail: raviprasadravuri2021@gmail.com
Received 30 October 2021; Accepted 24 December 2021; Publication 17 March 2022
Abstract
The objective of this paper is to explore the connotation among refractive error, intraocular pressure (IOP), and primary open-angle glaucoma (POAG) among the Wheatish populace. Population-based studies give significant data with respect to the predominance and hazard factors which include refractive error for glaucoma. The connotation between myopia and glaucoma is robust at lower IOP levels and deteriorated slowly with increasing IOP. This study shows the relationship between IOP and glaucoma is solid at a lower mean value of 15 3.23 levels and debilitated progressively with expanding IOP of 17.59 3.33 for PACG, 18.85 1.20 for POAG, 18.59 2.52 for PIGM and 19.12 1.42 for OH among Wheatish population. The illustration of glaucoma in myopic eyes deteriorated with growing IOP and no association has been detected with IOP 35 mmHg.
Keywords: Refraction error, populace-based study, POAG, Wheatish population, glaucoma.
1 Introduction
Refractive error and intraocular pressure (IOP) are two key risk factors for POAG, and refractive error is becoming more widespread over the world, indicating that it plays a substantial role in POAG improvement. The most common symptoms of refractive error include diplopia, haziness, glare or halos around bright lights, squinting, headaches, impaired vision, and eye strain (RE). Glaucoma is an unpredictably fatal disease that destroys the optic nerve and results in dynamic, permanent vision loss. It is the second most common cause of blindness worldwide [1, 2]. The visual field test, corneal thickness and point tests, optic nerve imaging, and an eye weight check are all examples of glaucoma eye tests. RE’s key involvement in the genesis of glaucoma is also important for glaucoma detection. Diabetic Retinopathy is the result of this syndrome.
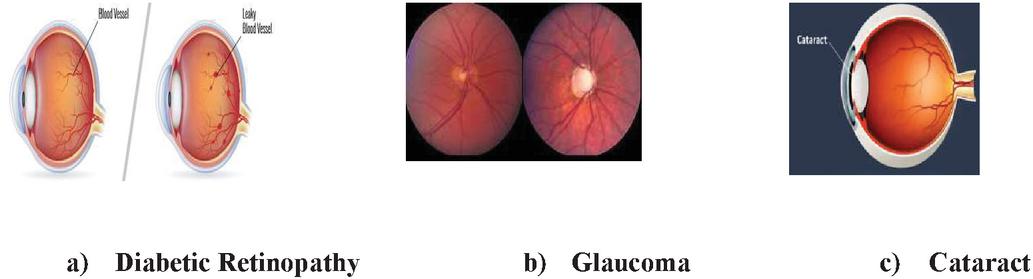
Figure 1 Chronic complications of diabetes in eye disease.
The unending entanglements of diabetes-related to eye infection is given in Figure 1. Among these chronic complications, Diabetic retinopathy is an important reason for blindness in Indian adults. In some human beings with diabetic retinopathy, retinal blood vessels may also swell and outflows fluid.
1.8 million (4.8%) of the 37 million individuals are visually impaired because of eye infections all through the world. Globally around 171 million diabetes patients are there, and it is expected to 366 million at the end of 2030. About half of the persons with diabetes are unconscious, albeit around 2 million died consistently are owing to the complexities of diabetes. In the upcomings 15 years, about 2% of people with diabetes may become daze, and about 10% may undergo serious visual misfortune. In the next 20 years, over 75% of patients are expected to have some type of diabetic retinopathy [3]. Glaucoma Research Foundation (GRF) conducted a review and discovered that 74% of quite 1,000 people met the aforementioned criteria and their eyes must be tested at regular intervals and 61% of those with enlarged eyes were identified to have glaucoma. 16% of African Americans were reported to have recently affected with glaucoma [4, 5]. Comprehensively, it is evaluated that around 1.31 billion individuals living with various types of vision disability. Additionally, 188.4 million individuals have a gentle vision disability, 216 million had modest to extreme vision hindrance, and 36 million people are visually impaired [6]. Regarding close vision, 827 million people living with a close vision weakness [7].
In this paper, a huge populace overview has been performed so as to distinguish patients with undetected glaucoma. The subsequent material, comprising of in excess of 4000 people among Wheatish populace. The reason for the present examination is to contemplate the connection among RE and glaucoma, all the more explicitly to see whether nearsightedness can be affirmed as a significant hazard factor for glaucoma, and the relationship among myopia and glaucoma depend on IOP.
This paper is arranged as follows. Section 2 exhibits a brief review of the literature. Section 3 defines the materials and methods. Section 4 gives the simulation results and findings, and Section 4 concludes the paper.
2 Review of Literature
Numerous elements have recently been accounted to influence IOP. A higher IOP has been associated with greater central corneal thickness [8], blood pressure [9], and body mass index [10]. It has also been stated that the existence of diabetes mellitus was a hazard factor for higher IOP. The investigations which cannot exhibit temporal causation, it is important to assess their relationship with IOP longitudinally. The relationship between RE and glaucoma subtypes with 95% confidence intervals measured as odds ratios (ORs). Myopia was related to an expanded occurrence of all types of POAG and OHTN, while hyperopia is related significantly to PACG occurrence. While higher myopia was a strong hazard factor for subtypes of glaucoma, low and medium myopia often affects the risk of glaucoma. Also, there were significant racial differences in the correlation of myopia with the danger of POAG and NTG [11].
In [12], researchers investigated the relationship between refractive errors to glaucoma and IOP in a given white population and found a cross-sectional relation of myopia with higher IOP and prevalent glaucoma. In [13], assessed refractive results of cataract surgery with intraocular lens (IOL) implantation according to primary congenital glaucoma (PCG). There were a greater prediction error and a greater myopic shift between PCG eyes after IOL implantation. Children’s eyes with PCG are more prone to refractive surprises because their changes in axial length are more delicate to IOP fluctuation.
A study has been conducted about the eyes that have undergone phacoemulsification with eyes that have not to experience glaucoma surgery at least 3 months after trabeculectomy and revealed that the adjustment in IOP was adversely related with refractive surprise; a connection was expected among lower postoperative IOP or greater reduction in IOP and reduction in axial length [14]. A report has uncovered that combined versus subsequent trabeculectomy, the refractive result was lower [15]. Then again, there were no differences in refractive and visual results in a study involving combined trabectome-cataract surgery with cataract-alone surgery, and a retrospective analysis demonstrated favorable refractive results in patients experiencing concurrent cataract removal with trabeculectomy or glaucoma drainage system surgery. Shanmugam et al. developed a method for detecting glaucoma in fundus images by measuring CDR (Cup to Disc Ratio) [16]. Raja et al. [17] developed a method for diagnosing glaucoma that focuses on the optic cup-to-disc ratio, which could be the optimum system for developing a capable, energetic, and accurate structure for glaucoma analysis.
There are few population-based studies have been directed outside the USA or UK that inspected the frequency of DR. In this examination, patients who were resolved to have diabetes from 1980 to 1984 had a 47% reduction in the risk of DR compared to patients analyzed from 1975 to 1979 [10]. The diminishing in risk was significantly more unmistakable in the associate broke down from 1985 onwards, at 64%. This assessment shows that while virtually all patients with type 1 diabetes may eventually make DR after some time, the frequency of type 1 diabetes patients with DR was undoubtedly on the decline. A population-based study between the Wheatish population-Diabetic individuals have been directed in this paper to discover the relationship between Refractive Error, IOP, and Glaucoma Etiology.
3 Materials and Methods
3.1 Recruitment
For this study, both men and women with the age of 40 are invited. An invitation number has been sent to every individual, offering a free eye health examination and an appointment. The invitation has been accompanied by a short questionnaire regarding eye history and ongoing medications. For this investigation, the two people with the age of 40 are welcomed. A greeting number has been sent to each person, offering a free eye wellbeing assessment and an arrangement.
3.2 Populace-based Study
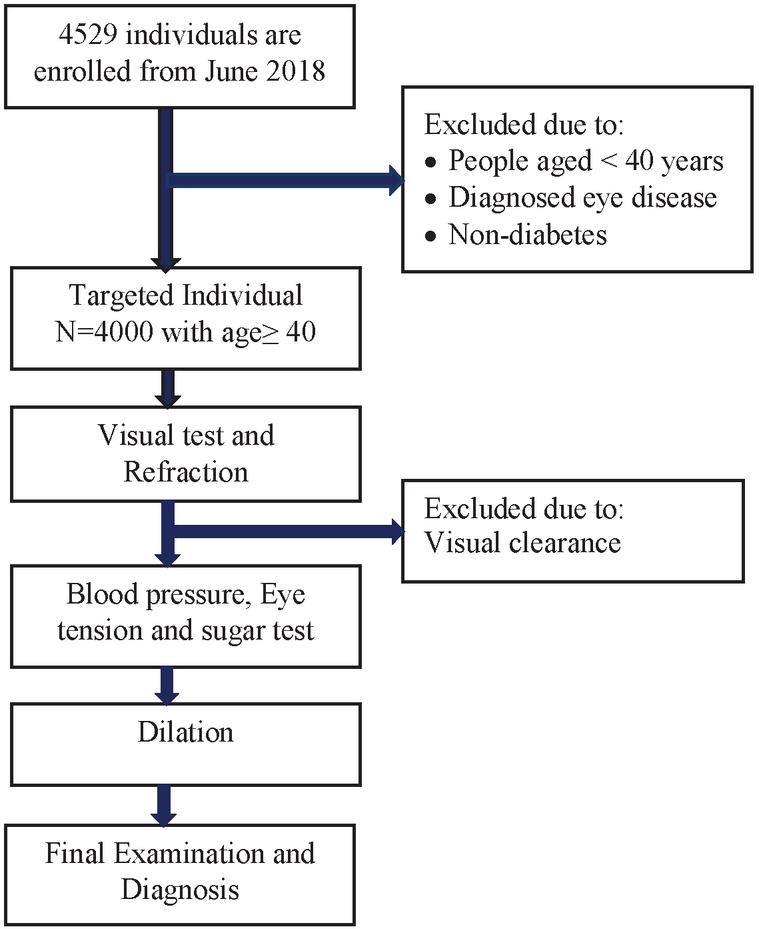
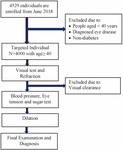
In this paper, the targeted individual number is 4000 with the age group of greater than 40 years with male and female diabetes. The workflow diagram of this study is given in Figure 2. Here, a totally of 4529 individuals have been investigated from 2018. In that 529 individuals have been excluded due to certain reasons which include people aged less than 40 years, some of the individuals are already diagnosed with their eye disease and are non-diabetic. Visual and refraction tests have been performed on the identified individuals. If the identified individuals have a clear vision, they are excluded from the study. Then the identified individuals are checked for blood pressure, eye tension and sugar test. Based on the testing results, final diagnosing has been performed for the individual. The examination procedure is as follows.
3.2.1 Vision test
Initially, every individual has gone through a vision test to identify the vision level. This test will help a doctor determine whether the individual vision has been affected by glaucoma. During this test, every individual is asked to look straight ahead and then indicate when a moving light passes your peripheral vision. At first, every individual has experienced a vision test to distinguish the vision level. This test will enable a specialist to decide if the individual vision has been influenced by glaucoma. During this test, each individual is approached to look straight ahead and afterward show when a moving light passes your fringe vision.
3.2.2 Refraction
A refractionist placed a series of lens in front of an individual’s eyes and asked which looks clear. This test determined the exact eye power and the level of sight. The blood pressure, intraocular pressure and a urine test are also carried out for diabetes and noted for further treatment.
Figure 2 Workflow diagram.
4 Dilation
In some cases, the refractionist instilled eye drops in the eye. It makes enlarge pupils bigger in order to get a better view of the inside of the eye. Dilation has taken 30 minutes of duration. At that time, they are asked to wait in the waiting room. The dilated individuals experienced a blurred vision for 2–3 hours after dilation.
5 Final Examination and Diagnosis
After all the tests have been carried out, the individuals are guided to the specialty clinics depending on the results.
6 Results and Discussion
The prevalence of glaucoma in screened subjects are tabulated in the Table 1. At the time of the screening examination, all participants are between 40 and 78 years of age. We identified 742 individuals with previously undiagnosed primary open-angle glaucoma (POAG), normal-tension glaucoma (NTG), primary angle-closure glaucoma, pigmentary and ocular hypertension (OH) corresponding to 18.55% of those screened. We found an additional 268 individuals with glaucoma who had been previously diagnosed and 23 individuals with other types of glaucoma.
Table 1 Prevalence of glaucoma in screened subjects
| N (Subjects) | % | |
| Patients with detected glaucoma | 742 | 18.55 |
| Patients with previously diagnosed glaucoma | 268 | 6.7 |
| Patients with secondary or angle-closure glaucoma | 23 | 0.57 |
| Normal | 2,967 | 74.17 |
| Total | 4,000 | 100 |
A total of 1,484 eyes are comprised in the present investigates, amongst them, 1,216 (81.94%) are glaucoma eyes. In that, 598 right eyes and 618 left eyes. We excluded 268 (18.05%) cases that are unable to obtain pictorial acuteness, visual clearance, and eyes that had before experienced cataract operation and the details are tabulated in Table 2.
Table 2 Number of eyes (both left and right) included in this study
| N (eyes) | % | |
| Total number of eyes screened | 1,484 | |
| Non-eligible | 268 | 18.05 |
| Eligible glaucoma eyes with IOP 25 mmHg | 785 | 52.89 |
| Eligible glaucoma eyes with IOP 25 mmHg | 431 | 29.04 |
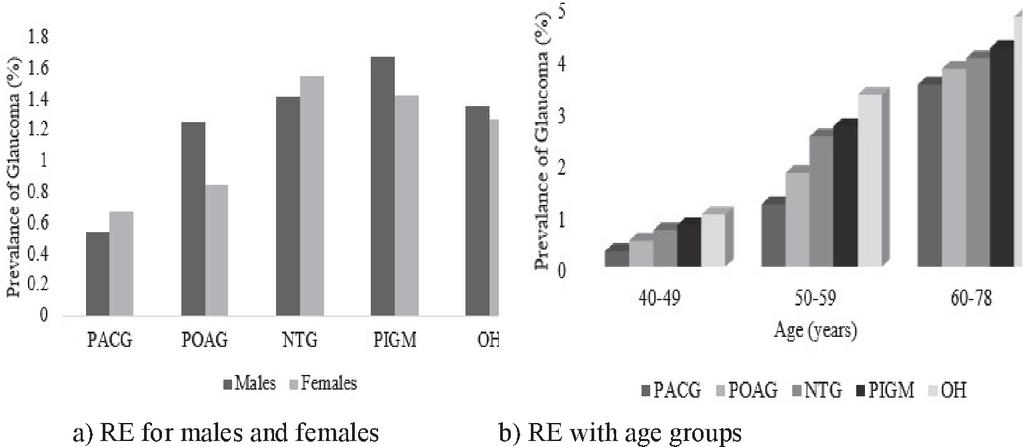
Figure 3 Prevalence of glaucoma in eyes.
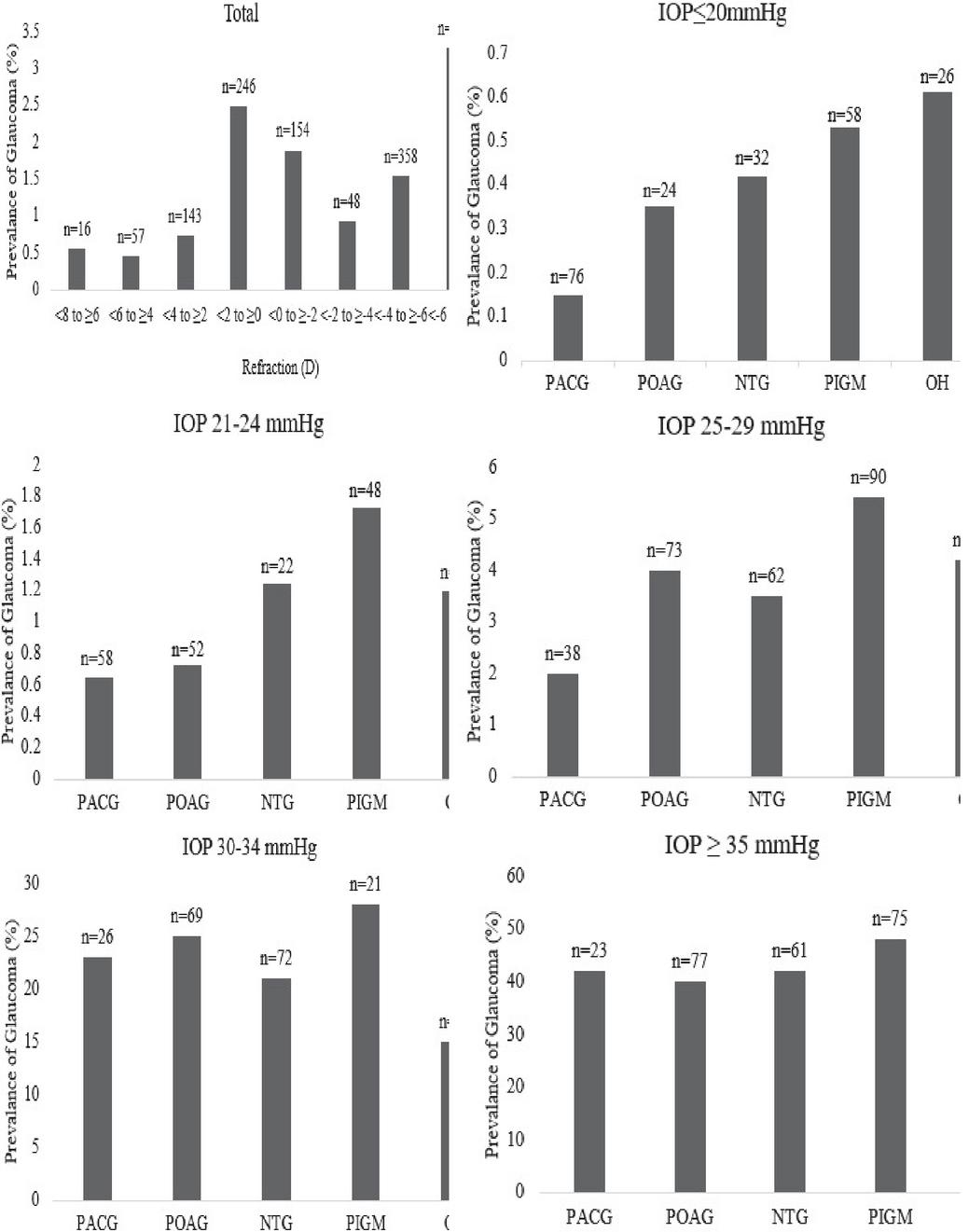
Figure 4 Prevalence of glaucoma in eyes with various RE and IOP-levels.
The connection among glaucoma and RE is detected in both males and females (Figure 3(a)), and across the age group is shown in Figure 3(b). We found a solid relationship between glaucoma and myopia, but also a strong reliance on IOP for the relationship. With rising IOP, glaucoma over-representation in the myopic group deteriorated slowly. There is a striking, practically straight connection among the occurrence of glaucoma in the group with IOP 20 mmHg (Figure 4) which occurred entirely in the eyes with IOP 35 mmHg and this distinction shows the huge connection among IOP and RE. The association is good at minor IOP levels and completely vanished at higher IOP levels indicates that for normal stress glaucoma, myopia was an important risk factor.
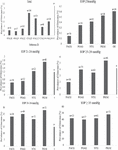
Table 3 gives the features of the 5-glaucoma case and non-glaucoma controls. Glaucoma patients with 40 years with diabetic patients have been considered for this trial. Ladies set up 62% of controls, 71% of PACG patients, 56% of POAG, 49% of PIGM and 69% of OH with diabetic patients. For men 59% of controls, 37% of PACG patients, 56% of POAG, 62% of PIGM and 44% of OH independently. The regularity of glaucoma subclasses are moved by race and ethnicity. Clinical incorporates moreover fluctuated with the subclasses of glaucoma. True to form, glaucoma cases commonly greater CDRs and greater IOPs than controls. Similarly, the diabetics have a higher estimation of IOP, lower estimation of CDR, and higher estimation of RE, however, various patients had respectably lower estimation of IOP, higher estimation of CDR, and lower estimation of RE.
In India, 4.042 million with the age gathering of 40–49 years and 30.970 million with more noteworthy than 50 years are influenced by uncorrected refractive error [16]. The average IOP is 15.61 3.09 mm Hg for women and 15.54 3.54 mm Hg for men separately [14]. IOP for women and men is comparative within the same age group, while IOP in earlier age is greater when contrasted with older-age groups. The mean IOP for the diabetic populace is 15.08 2.18 mm Hg.
The Spherical Equivalent (SE) refractive erroris determined as the circle 1/2 chamber. The SE is calculated by using the following equation,
| (1) |
With a spectacle amendment of , the . Nearsightedness has been characterized using SE as 0.50 diopters (D); mellow nearsightedness is characterized as 3.0 D; moderate nearsightedness is characterized as 3.0D, and high nearsightedness is characterized as 6.0D. Mean IOP has been found that 15.98 mmHg in normal hyperopic eyes and increased gradually with increasing myopia to 16.2 mmHg. The normal and glaucomatous eyes related to refractive error and IOP have been listed in Table 4.
Table 3 Features of Glaucoma and controls for Wheatish population
| Controls | PACG | POAG | NTG | PIGM | OH | |
| Gender | ||||||
| Female | 62.25 | 71.23 | 56.34 | 58.69 | 49.64 | 69.48 |
| Male | 58.95 | 36.92 | 56.34 | 48.64 | 62.76 | 44.68 |
| Age | ||||||
| 40–49 | 27.30 | 4.29 | 6.14 | 5.89 | 13.79 | 11.32 |
| 50–59 | 40.10 | 28.36 | 31.40 | 32.59 | 43.69 | 40.69 |
| 60–78 | 32.50 | 65.89 | 62.50 | 62.25 | 45.95 | 51.69 |
| Mean SD | 57.28 13.5 | 67.27 10.65 | 68.51 10.38 | 67.589.68 | 63.89 12.21 | 65.5813.50 |
| CDR | ||||||
| Mean SD | 0.31 0.13 | 0.45 0.11 | 0.56 0.23 | 0.64 0.19 | 0.51 0.25 | 0.45 0.36 |
| IOP | ||||||
| Mean SD | 15.98 3.23 | 17.59 3.33 | 18.85 1.20 | 14.58 3.273 | 18.59 2.52 | 19.12 1.42 |
Table 4 Mean IOP in normal and glaucomatous related to RE
| Normal Group | Glaucoma Group | |||
| Mean IOP (mmHg) | N | Mean IOP (mmHg) | N | |
| Myopia (SE –0.50 D) | 15.6 | 1823 | 23.6 | 167 |
| Slight Myopia (SE –3.0 D) | 15.9 | 1526 | 21.3 | 256 |
| Moderate Myopia (SE –3.0 D) | 16.4 | 532 | 22.1 | 92 |
| High Myopia (SE –6.0 D) | 16.9 | 425 | 21.2 | 45 |
Therefore, the examination of this huge populace-based study indicated that the incidence of glaucoma is significantly reliant on RE and signified in bigoted eyes. The connection among glaucoma is stronger in eyes with low IOP, weaker in the intermediate pressure readings group, and vanished in eyes with high pressure.
7 Conclusion
Populace based health examination has been directed for diabetes in India. Increasingly serious nearsightedness is related to a more prominent probability of glaucoma in a Korean population matured 40 years or more. This constructive connection among the RE and IOP underlines the significance of glaucoma observation in the nearsighted populace in light of the fact, that both raised IOP and nearsightedness are significant hazard factors for glaucoma development. This paper demonstrates that there is a solid association among RE, IOP, visual biometry and POAG for diabetes more protuberant than 40 years in the Wheatish populace. The incorrect RE can be rectified with eyeglasses while cataracts medical procedure can reestablish image. Vision recovery is additionally powerful in enlightening irretrievable vision debilitation. In this study, it is found that the individual group with the age of 60–69 is more affected by glaucoma with 65.89% of PACG, 62.50% of POAG, 62.25% of NTG, 45.95% of PIGM and 51.69% of OH among Wheatish population. Curiously the relationship among refractive error and IOP varied by age group, yet IOP is unequivocally connected with glaucoma and refractive error for diabetes.
References
[1] Resnikoff S, Pascolini D, Etya’ale D, Kocur I, Pararajasegaram R, Pokharel GP, et al. Global data on visual impairment in the year 2002. Bull World Health Organ 2004; 82:844–51.
[2] Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol 2006; 90:262–7.
[3] Global initiative for the elimination of avoidable blindness: action plan 2006–2011, Vision 2020, World health organization, pp. 1–97.
[4] Resnikoff, Donatella Pascolini, Silvio P Mariottiand Gopal P Pokharel. Global magnitude of visual impairment caused by uncorrected refractive errors in 2004, Bulletin of the World Health Organization January 2008, 86(1) 63–70.
[5] George R, Ramesh S Ve, and Viajaya L. Glaucoma in India: Estimated burden of disease. J Glaucoma 2010; 19:391–7.
[6] Bourne RRA, Flaxman SR, Braithwaite T, Cicinelli MV, Das A, Jonas JB, et al.; Vision Loss Expert Group. Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: a systematic review and meta-analysis. Lancet Glob Health. 2017 Sep;5(9): e888–97.
[7] Fricke, TR, Tahhan N, Resnikoff S, Papas E, Burnett A, Suit MH, Naduvilath T, Naidoo K, Global Prevalence of Presbyopia and Vision Impairment from Uncorrected Presbyopia: Systematic Review, Meta-analysis, and Modelling, Ophthalmology. 2018 May 9.
[8] Foster PJ et al., Intraocular pressure and corneal biomechanics in an adult British population: the EPIC-Norfolk eye study. Invest Ophthalmol Vis Sci 2011; 52: 8179–85.
[9] Yokomichi H, Kashiwagi K, Kitamura K et al. Evaluation of the associations between changes in intraocular pressure and metabolic syndrome parameters: a retrospective cohort study in Japan. BMJ Open 2016; 6: e010360.
[10] Jang HD, Kim DH, Han K et al. Relationship between intraocular pressure and parameters of obesity in Korean adults: the 2008–2010 Korea National Health and Nutrition Examination Survey. Curr Eye Res 2015; 40: 1008–17.
[11] Ling Shen, et al., The Association of Refractive Error with Glaucoma in a Multiethnic Population, Ophthalmology 2015; pp. 1–10.
[12] Tien Yin et al., Glaucoma in a white population, American academy of ophthalmology, 2003, pp. 211–217.
[13] Sudarshan Khokhar et al., Refractive outcomes of cataract surgery in primary congenital glaucoma, eye, 2018.
[14] Tedja MS, Wojciechowski R, Hysi PG, et al. Genome-wide association meta-analysis highlights light-induced signaling as a driver for refractive error. Nat Genet. 2018; 50:834–848.
[15] Thorleifsson G, Walters GB, Hewitt AW, et al. Common variants near CAV1 and CAV2 are associated with primary open-angle glaucoma. Nat Genet. 2010; 42:906–909.
[16] P., Shanmugam, Raja J., and Pitchai R. “An Automatic Recognition of Glaucoma in Fundus Images Using Deep Learning and Random Forest Classifier.” Applied Soft Computing 109 (2021): 107512.
[17] Raja, J., P. Shanmugam, and R. Pitchai. “An Automated Early Detection of Glaucoma using Support Vector Machine Based Visual Geometry Group 19 (VGG-19) Convolutional Neural Network.” Wireless Personal Communications 118.1 (2021): 523–534.
Biography

Ravi Prasad Ravuri working as an Application Developer for Sriven Technologies. His current research focus is on Software Quality, Cloud Computing and securing Cloud environments. He holds his B.Sc degree in Statistics & Computer Science from Kakatiya University, India in 1995 and M.C.A from Osmania University in 1998, M.Tech in Computer Science at Osmania University, Hyderabad in 2006, and completed Ph.D. in Software Quality at Nagarjuna University Guntur. He has organized no. of workshops & technical symposiums in various Computer Science related fields and published papers in various journals.
Journal of Mobile Multimedia, Vol. 18_4, 1177–1190.
doi: 10.13052/jmm1550-4646.18410
© 2022 River Publishers