Deep Learning Methods for Lung Cancer Nodule Classification: A Survey
Pavan Kumar Illa1, 2,*, T. Senthil Kumar1 and F. Syed Anwar Hussainy1
1Department of Computer Science and Engineering, SRM Institute of Science and Technology, India
2Department of Information Technology, VNRVJIET, Hyderabad, India
E-mail: illa.pavankumar@gmail.com
*Corresponding Author
Received 23 June 2021; Accepted 01 September 2021; Publication 29 October 2021
Abstract
Lung cancer is one of the leading causes of cancer related deaths. It is due to the complexity of early detection of nodules. In clinical practice, radiologists find it difficult to determine whether a condition is normal or abnormal by manually analysing CT scan or X-ray images for nodule identification. Currently, various deep learning techniques have been developed to identify lung nodules as benign or malignant, but each technique has its own advantages and drawbacks. This work presents a thorough analysis based on segmentation techniques, Related features-based detection, multi-step detection, automatic detection, and deep convolutional neural network techniques. Performance comparison was conducted on a selected works based on performance measures. A potential research direction for the recognition of lung nodules is given at the end of this study.
Keywords: Lung cancer, lung nodules, segmentation, deep neural network, feature extraction, automated detection.
1 Introduction
Lung cancer is considered as the lethal cancer in the world and hence, various nations are designing policies for early diagnosis of lung cancer. In general, lung cancer is spotted based on the existence of lung nodules. They are tiny multitudes of tissue in the lungs. Cancer nodules are described as malignant. The size of malignant nodules is greater than that of healthy nodules. Special attention is required for nodules with a diameter greater than that of a small. Computer tomography (CT) scans are generally used to identify lung nodules. A CT scan helps to diagnose lung cancer at the initial stage and supports to control lung cancer consistently at a later stage. Computer-aided detection (CAD) enables radiologists to determine the size and expansion of the nodules easily and reliably. During follow-up monitoring, CAD systems will assist to detect the increase in size of nodules and the presence of new nodules.
Regrettably, lung cancer signs do not manifest until late in the disease’s course, hence it is difficult to treat and not curative. Various imaging modalities such as chest X-rays, CT scans, positron emission tomography (PET) scans and magnetic resonance imaging (MRI) can be employed to detect and discover the severity levels of lung cancer. Low-dose CT scans (LDCT) are being utilized for screening of individuals to find the chances of acquiring lung cancer in the future. In the year 2011, the “National Lung Screening Trial (NLST) [1] research team study results indicate that the patients who underwent low-dose helical CT scans had a 15% to 20% lower chance of dying of lung cancer than those who underwent normal chest X-rays”.
2 Deep Learning
Deep Learning (DL) is an efficient and effective machine Learning (ML) approach that employs a variety of neural network models to perform a variety of imaging tasks, including segmentation, object recognition and classification. Traditional machine learning methods depend on feature extraction techniques to train the algorithm, whereas deep learning models explicitly study the image data without any need for Feature Extraction (FE). Till this day, many deep learning models are in operation, which include: Recurrent Neural Network (RNN) [2], Convolution Neural Network (CNN) [3], Auto encoder [4], Generative Adversarial Network (GAN) [5], “Restricted Boltzmann Machines” (RBMs) [6], “Deep Belief Networks”(DBN) [6], Long Short Term Memory Networks (LSTMs) [7], Radial Basis Function Networks (RBFNs) [8–10], Multilayer Perceptron’s (MLPs) [11], Self-Organizing Maps (SOMs) [12].
3 Lung Nodule Classification with Deep Learning
Early detection of a nodule will increase a patient’s odds of survival. Identifying the stages of cancer will also aid in the delivery of adequate therapy to patients. Analysing the diagnostic images is a difficult task for radiologists because CT images have a high dimensionality, and the nodule may be thin. As a result, computer aided diagnostic systems are required. Computerised detection of lung cancer helps the radiologist to take correct decisions.
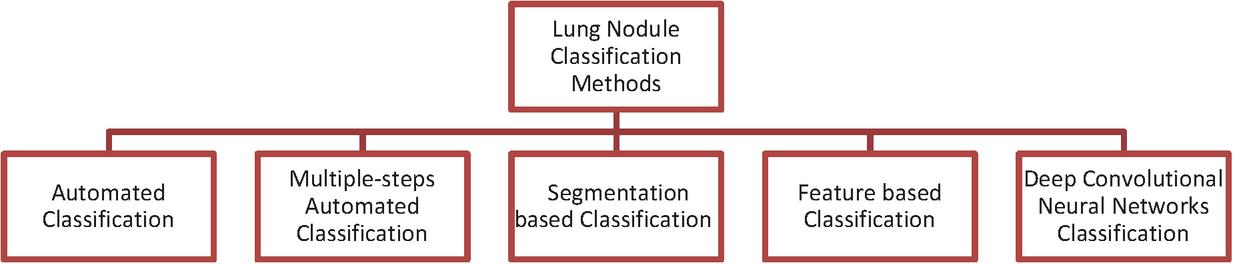
Currently, Deep Learning is used extensively for medical image processing. Deep Learning technology dominates the traditional ML techniques in classifying the images. In recent years, many different DL methods were developed for lung cancer nodule identification and obtained better classification accuracy rates. In this paper, recently developed deep learning methods were analysed and examined in detail, highlighting their advantages and drawbacks. Based on their execution, we divided these approaches into five groups as shown in Figure 1. The first group includes automated classification systems. The automatic classification was performed in several stages by the second group of methods. The third group focuses on segmentation. The fourth category focuses on features. The fifth group is concerned with the implementation of Deep Convolutional networks. In subsequent sections we presented a detailed review of the methods in these five groups along with their advantages and drawbacks.
Figure 1 Categories of lung nodule classification methods.
3.1 Automated Lung Nodule Detection Methods
This section includes a study on automated classification systems.
Saba et al. [13] instigated an automatic technique to find lung nodules class as cancer or non-cancer. This method includes segmentation, lesion intensification in conjunction with feature extraction for every individual lesion. This method used a blended mechanism of multi-classifiers logistic regression, multi-layer perceptron in conjunction with voted perceptron for categorizing Lung Nodule (LN) with k-fold cross-validation(k 3,4,5). It was validated using open-source data set “Lung Image Database Consortium” (LIDC) and found that the method surpassed the existing top-class techniques with an accuracy rate of 100%.
Teramoto et al. [14] introduced a hybrid method for finding lung nodules using PET/CT images. The CT image first detected a mass region based on a “cylindrical nodule enhancement filter (CNEF)” generated by a contrast improving kernel. High-uptake areas were spotted with PET images and were later combined with the CT image detected area. False positive (FP) characteristics from PET and CT images were eradicated by a rule-centred classifier and by Three Support vector machines (SVM). The detection capacity was experimentally tested based on 100 PET/CT pictures cases. As a result, 83 percent of candidates were responsive with 5 FPs/case.
Shen et al. [15] suggested parameter-free lung segmentation. to increase the identification accuracy of juxta pleural nodules in the lung. By employing a two-way chain coded method, the lung border was smoothened along with a support vector machine. It was validated using the open-source data set Lung Image Database Consortium (LIDC) on 233 computed tomography studies representing 403 juxta pleural nodules. The re-inclusion rate of the strategy was 92.6%. The accuracy of segments was additionally validated with the 10 arbitrarily selected CT sequence and attained 0.3% average over-segmentation along with 2.4% under-segmentation rates, which is contrasted with the norms set by specialists.
Naqi et al. [16] introduced a hybrid model to identify nodules and their class. Firstly, an optimum grey level threshold was used to extract the lung area. Subsequently a hybrid feature vector was designed by fusing geometric structure and “Histogram of Oriented Gradient reduced by Principal Component Analysis (HOGPCA)” feature. Upon extraction, classification was done by four classifiers: k-Nearest Neighbourhood (k-NN), SVM, Naïve
Bayesian in conjunction with AdaBoost. It was validated using publicly available data set Lung Image Database Consortium (LIDC). Accuracy rate of 99.2% has been achieved with AdaBoost classifier. Zhang et al. [17] suggested that the vesselness filter could provide a strong pulmonary recognition method with “Multi-Scene Deep Learning Framework (MSDLF)”. To improve the perception of radiologist, four-channel CNN model is devised with 4-stage nodules that integrated dual image scenes. Two separate classes applied the method. From he results, it is evident that the MSDLF was successful in improving the exactness of lung nodules, while reducing the false positives in huge quantities of image data.
Table 1 illustrates the various lung nodule detection techniques based on automated classification with respect to their advantages and drawbacks.
3.2 Multiple Steps Automated Classification for Lung Nodule Detection
This section includes a study on the automatic classification methods carried out in several stages.
Zuo et al. [30] developed a multiple-resolution CNN. It extracted features of disparate levels together with resolutions from distinct depth layers on the network intended for nodule classification. First, it transferred knowledge from the CNN which implemented to detect edges as well as to enhance the model to a multiple-resolution model that was appropriate for the image classification task. The model was validated with LUNA16 data set, and it was shown that the method has a precision of 0.9733, an accuracy of 0.9673 and a curve area of 0.9954 higher than the values obtained from most of the advanced approach, but the technique is poorly accurate for large quantities of data.
Saba et al. [24] depicted an approach that identified the lung nodule at the preliminary stage. This method was executed in three major phases: (i) Nodule segmentation centred upon Otsu threshold; (ii) extraction of geometrical, texture along with DL aspects for choosing optimum features; (iii) The optimum features were combined serially for Nodule categorization into malignant or benign. It was validated using the open-source data set “Lung Image Database Consortium (LIDC)”. The approach Contrasted with the prevailing methods by exhibiting better performance.
Table 1 Automated classification methods
| Reference | Technique | Advantages | Drawbacks |
| Saba et al. [13] | Lung nodule identification by applying multiple classifier voting | It exhibited 100% Accuracy | – |
| Teramoto et al. [14] | Hybrid method using both CT and PET images for nodule detection | Exhibiting better performance in detecting lung nodules in mass-screening settings | Detection capability can be further improved |
| Shen et al. [15] | Parameter-free Lung segmentation Algorithm | Exhibiting better performance in handling small images with low image quality. | All Juxta-pleural nodules are not handled. |
| Naqi et al. [16] | Hybrid 3D-nodule detection method (after identifying the lung area) Geometric Texture HOG PCA | accuracy of 99.2% has been achieved | High Computational time. |
| Zhang et al. [17] | MSDLF utilizing Vesselness Filter. | Incredible reduction on False positives. | – |
| Zheng et al. [18] | Maximum Intensity Projection – Based convolution Neural Network (MIP-CNNs) | it detected nodules of volume 3 mm–10 mm and ensured less false positives | – |
| Huang et al. [19] | noisy U-Net (NU-Net) | uncertainty in the output result caused by noise has been removed | There aren’t enough features to capture all the intricate properties of lung nodules. |
| Ling et al. [20] | CNN-SVM | Low computational time, sufficient features were used to classify different kind of lung nodules. | Accuracy was low and leaded to misclassification |
| Shen et al. [21] | Multi-Crop CNN | low time consumption that avoided the unnecessary time on feature extraction and pre-processing | Highly addicted to noise, Misclassification, highly challengeable to locate the location of the nodule. |
| Jiang et al. [22] | Four channel CNN | Handled huge data, Low False Positive Rate (FPR) | Low Reliability |
Table 2 Automated classification methods with multiple steps
| Reference | Technique | Advantages | Drawbacks |
| Zuo et al. [23] | Multi-resolution CNN +knowledge transfer | Radiological heterogeneity identified successfully | lack of ability to grasp contextual information may lead to mis classification. |
| Saba et al. [24] | Ostu method (Morphological operations to segments) +PCA | Reduced False Positives | segmentation based results are not promising. |
| Merja et al. [25] | Ostu method +Semantic Segmentation | Reduction in False Positive rate (FPR) and better Sensitivity rate. | – |
| Patrice et al. [27] | Multiple-view 3D-CNN’s ELM | Small size lumps detected | Fusion strategy to be selected with caution. may lead to poor performance. |
| Tan et al. [28] | Segmentation +Classification based on 3D-CNN | High sensitivity for identification of cancer nodules | – |
| Zheng et al. [29] | CNN Artificial Immune Ensemble Algorithm | Results of Sensitivity and accuracy were promising | Performance measures can be improved further |
| Yongqiang Tan et al. [30] | Morphological dot-enhancement regression tree classifier | Partition of candidate nodules is easy | Need to improve weak nodes |
| Wang et al. [31] | Multilevel feature extraction DeepLN | Recognize lung nodules in low- and high-resolution CT images. | It requires a significant amount of computational power. |
| Imdad et al. [32] | EL transferable texture Convolutional Neural Networks | Reduced Computational Time & complexity to detect nodules | – |
| Kuo et al. [33] | Adaptive wiener filter +Otsu method+ feature extraction SVM | detected “ground glass opacity (GGO), part solid, and solid nodules” in CT. | – |
Meraj et al. [25] established a system for identification and classification of nodules as cancer or non-cancer. This system has been evaluated based on the public data set, i.e., image collection from the “Lung Image Database Consortium (LIDC)”. OTSU and semantic segmentation adaptive threshold techniques were used for detecting unhealthy lung nodules precisely. Overall, principal component analysis algorithm was used to retrieve 13 nodules. Empirical research demonstrated that the method surpassed other technologies and gave 99.23% accuracy with logit boost classification.
Yuyun Ye et al. [26] developed an automated nodule detection model centred upon the modified V-Nets (aimed at nodule candidate detection) and higher-level descriptor centred SVM (aimed at FP reduction). To ameliorate the FP diminution’s performance, hard mining intended for retraining was executed. The SVM functioned better in FP diminution, and it employed more critical aspects of CT. To exhibit the CAD’s effectiveness, it was validated on LIDC data set.
Patrice et al. [27] introduced a system to distinguish between the micro-nodules and non-nodules on CT images through ensemble learning. ‘5’ disparate 3D-CNN were constructed and executed on one size of the nodule candidates. To incorporate five 3D-CNN outputs, an Extreme Learning Machine (ELM) was employed and yielded the last classification outcomes. The system performance was assessed by accuracy, F-score, AUC, as well as sensitivity. The system attainted 97.35% accuracy, 0.98 AUC, 96.42% F-score, and 96.57% sensitivity, respectively. Table 2 illustrates the various lung nodule detection techniques based on automated classification carried-out in multiple steps with respect to their advantages and drawbacks.
3.3 Segmentation Based Classification for Lung Nodule Detection
This section contains a literature review on the third category of DL methods focused on segmentation.
Naqi et al. [34] designed a multistage segmentation model. The lung region was mined using a combination of “corner-seeded region growing” and “differential evolution-based optimal thresholding”. To smoothen boundary lines, fill spaces, and delete nodules, morphological operations were employed. Geometric features as well as 3D edge information were employed to extract nodule candidates. The categorization was performed over features derived from Geometric texture features descriptor (GTFD) and the results showed an accuracy rate of 99%, sensitivity measure of 98.6%, precision of 98.2%, and FPR of 3.4.
Feng et al. [35] developed mechanisms for the incorporation of nodule detection and segmentation into a blend of “SLIC super voxel segmentation and CNN classification”. CNN learning requires only loosely labelled data. In this case, CNN needs only a single label for each annotated object. The CNN architecture was designed as a three-dimensional multi-level structure capable of identifying and clustering nodules of various shapes and dimensions. An experiment on the LUNA16 dataset demonstrated prominent performance and demonstrated the need and efficiency of multi-level neural network design and multi-stage system for computer assisted detection of pulmonic nodules.
Centred on multilevel thresholding, John et al. [36] suggested a method for pulmonic nodule segmentation and FE. The features derived can help to classify pulmonary nodules effectively. There was heavy segmentation with three stage thresholding in addition to the derived functionality, which resulted in less false positives for any suitable classification architecture.
Nithila et al. [37] developed a “region-based active contour model” and the “Fuzzy C-Means (FCM) technique”. CT photographs acquisition, lung parenchyma reconstruction together with segmenting LN is the major intent of this work. Selective binary, and Gaussian filtering with new signed pressure force function were employed for LN segmentation and clustering technique was used to reconstruct the parenchyma.
For nodule candidate segmentation and FPs reduction, Moghaddam et al. [38] established a new hybrid system. The photos were first relocated to the neutrosophic domain. The performance of the previous step was then filtered out using three filters: Blob-like Structure Enhancement (BSE), Line Structure Enhancement (LSE) in conjunction with Central Adaptive Medialness (CAM) filters. These filters outcomes were utilized for nodule detection and segmentation, respectively. A candidate voxel line tracking system is created and then used for LN segmentation. An approach called Sparse Coding was implemented to learn the function vector after FE. In the final stage, the data was classified using the generalised linear regression model. The classifier’s output for sensitivity is 98.32% and FP/scan is 2.8, but the process failed to detect a significant number of nodules.
Table 3 illustrates the various lung nodule detection techniques based on segmentation of nodules along with the segmented region with advantages and drawbacks.
Table 3 Segmentation based classification methods
| Reference | Technique | Segmented Region | Advantages | Reference |
| Naqi et al. [34] | Hybrid deep learning model with GTFDSVM Ensemble classification | Multi segmentation model | Good at handling small nodules | may lead to Over segmentation |
| Feng et al. [35] | SLIC super voxel segmentation CNN classification | SLIC super voxel segmentation | Efficient in generating refined shapes of nodules | may lead to overfitting |
| John et al. [36] | 3-level segmentation model (Global Intermediate Moceo-level] | Segmentation using multilevel thresholding | Suitable for isolated nodules | Accuracy of the model was not tested, and Juxta pleural nodules are not covered |
| Nithila et al. [37] | Region based Active Contour+FCM+Binary and Gaussian filtering | Region based segmentation | Error rate minimized and similarity measure improved with low time complexity | Not mentioned about Specificity, Sensitivity and FP rate. |
| Moghaddam et al. [38] | Hybrid model (BSE LSE CAM filters] | segmentation blood vessels and pulmonary nodules | Sensitivity is improved and least FPs/scan value | Juxta-pleural nodules need to be addressed. |
| Ren et al. [39] | A semi-automated pulmonic nodule segmentation technique with an expanding region | Pulmonary Nodule Segmentation | False Positive Reduction and Sensitivity | The database’s nodule sizes were too tiny, resulting in incorrect classification results. |
| Rocha et al. [40] | SegU-Net | Segmented juxta-pleural nodules | Capable of detecting LN of diversity shape, size, texture in tandem with the existence of adjacent structures | Difficult for variety of LN as well as the visual aspects similarity between LN and their surroundings |
| Liu et al. [41] | OTSU’s curve thresholding segmentation method in two dimensions with Random Forest method | Pulmonic Parenchyma Segmentation | Capable to detect accurate nodule and avoided misclassification | Time complexity and loss minimization was complex process |
| Veronica et al. [42] | Fuzzy C-Means (FCM) ANN | Segmentation of lungs | Low structural complexity, low computational time | Inaccurate detection of nodule, sensible to noise |
| Haichao et al. [43] | DB-ResNet | Segmentation of Lung nodule of different categories | Detected LN’s of distinct kinds and the similarity of visual traits between LN’s and their nearby regions was also detected. | Image clarity was not proper that led to misclassification of nodules. |
3.4 Feature Based Classification for Lung Nodule Detection
Taşcı et al. [44] created a tool and system for automatically recognising a juxta-pleural nodule pattern in cross-sectional CT lung photographs. The system’s six main stages were pre-processing, segmentation, determining nodule candidate areas, FE, Feature Selection (FS) along with classification. It was based on the LIDC dataset, which included cross-sectional lung CT images for 24 patients. Total 1410 LN regions, and 40 features derived from 138 cross-sectional images. Ten classifier experiments were conducted, and the results were provided.
Raja et al. [45] proposed an innovative system for LN recognition established on a hybrid collection of features and an ANN. Firstly, using optimal thresholding, the lung region was segmented from the input CT picture. The lung area was segmented from the input CT image by employing “optimal Thresholding”. Later, for image enhancement filtering technique Multi scale dot augmentation was applied. Following that, LN candidates were identified an and Shape, Texture, Intensity features were extracted from the enhanced picture. Finally, a two-layer feed forward neural network was used to identify lung nodules. It was validated using the open-source data set “Lung Image Database Consortium (LIDC)”, which had a sensitivity value 95.5 percent with just 5.72 FP/scan.
Naqi et al. [46] developed a four-step nodule identification and categorization method. Firstly, this method extracts the lung area using optimal grey-level threshold determined by the “Darwinian particle swarm optimization fraction”. The LN recognition approach was established using Geometric fit in parametric form, where the geometric features of the nodules were incorporated. To properly describe the potential nodules, “hybrid geometric texture character description” was created afterwards. Finally, stacked auto-encoder neural network with softmax intended for feature reduction as well as classification was implemented. It was validated using the open-source data set “Lung Image Database Consortium (LIDC)” and the findings revealed that the approach had substantially lessened the frequency of false positives at the same time it is also being highly complex.
To minimise false positive effects, Wang et al. [47] built a deep feature fusion from non-medical training and hand-crafted features. Based on the findings of the public dataset experimentation, the deep fusion feature had sensitivity 69.3% and specificity value of 96.2 percent, respectively with 1.19 false positive per image, compared to single hand-crafted features 62 percent and 95.4 percent, respectively with 1.45 false positive/image.
da Silva et al. [48] demonstrated a deep learning model with an “evolutionary technique”, for reducing the FP number. To increase network efficiency and eliminate the need for manual investigation, the “Particle swarm optimization (PSO) algorithm” was used to boost the network hyper parameters in CNN. It was validated using the open-source data set “Lung Image Database Consortium (LIDC)” with a 97.62 percent accuracy, 92.20 percent sensitivity, 98.64 percent precision, and a 0.955 region under the receiver operating characteristic (ROC) curve.
Shaukat et al. [49] projected a method that includes pre-processing, noise drop from input image, and LN segmentation by means of optimal thresholding. Using multi-scale enhancement, the image was enhanced to identify the nodules. The classifier was able to accurately identify lung nodules with 95% accuracy. To maximise the sensitivity and minimise false positives, Texture, Shape and Intensity features were selected. Several other supervised classifiers like “K-Nearest-Neighbour (KNN)”, “Decision Tree” and “Linear Discriminate Analysis (LDA)” have also been compared. The classifier developed could not classify both the texts and morphological characteristics.
Table 4 illustrates the lung nodule detection based on various extracted features and the respective techniques along with their advantage and disadvantage.
Table 4 Lung nodule detection techniques based on features
| Reference | Technique | Feature Extracted | Advantages | Drawbacks |
| Taşcı et al. [44] | Deep learning model to detect Juxta pleural nodules with GLMR classifier | Shape and texture-based features (First and second order statistical features) | Better AUC value and reduction of false positives | only Juxta pleural odes were addressed. |
| Raja et al. [45] | Marker controlled watershed technique with Artificial Neural Network (ANN) classifier. | Texture Shape Intensity | False positive reduction and sensitivity improved | need to be tested on larger datasets |
| Naqi et al. [46] | Autoencoder +SoftMax classifier with fractional order Darwinian Practice swarm optimization | Texture and geometric features (2D and 3D). A hybrid vector is created | Significant reduction in false positives and promising sensitivity | Time complexity w.r.t training is more. |
| Wang et al. [47] | Deep feature fusion model with transfer learning | hand-crafted features | High accuracy and low FP rate | need to be tested on larger datasets |
| da silva et al. [48] | CNN Particle swarm Optimization | Intensity, shape, and geometry | AUC was improved | need to more robust and generic |
| Shaukat et al. [49] | Optimized feature set with SVM classifier | intensity, shape, and texture | significant reduction in FP rate | micro nodules need to be detected |
| Yutong et al. [50] | Deep Convolutional Neural Network (DCNN) with AdaBoost Back propagation neural network (BPNN) | Gray Level Co-occurrence Matrix (GLCM)-based texture descriptor, “Fourier shape descriptor to characterize the heterogeneity of nodules” | Better reliability, computational complexity was less | Irrelevant selection of the features |
| Yuan et al. [51] | Scale Invariant Feature Transform (SIFT)+Multiple Kernel Learning (MKL)+ Multi-View Multi-Scale CNNs | Statistical Features, Geometrical Features | Accuracy was improved to detect the lung nodule | Leaded to curse of dimensionality |
| Farahani et al. [52] | MLPs KNN SVM | Statistical and Morphological Features | False positive prediction was avoided | Leaded to over fitting of the data |
| Qin et al. [53] | Conditional Generative Adversarial Network (CGAN) | Semantic labels were produced to provide spatial contextual information to the network | Small nodules were detected | Highly complex, more computational time |
| Dhara et al. [54] | SVM | Features centred upon Shape, Margin and Texture. | The detection of nodules is simple to carry out. | For huge data it was highly complex |
| Shen et al. [55] | Stationary Wavelet Transform+ AdaBoost | a solitary feature, texture features | Majority based classification of lung nodules improved the accuracy | Clear illustration of the image was not obtained as it was literally based on frequency domain |
| Khehrah et al. [56] | SVM | Statistical and Shape-based Features | Extracted features improved the performance of the model | Irrelevant data caused misclassification of nodule |
3.5 Deep Convolutional Neural Networks Nodule Detection
This section includes a study on the methods concerned with the efficient implementation of Deep Convolutional networks.
Nasrullah et al. [57] instigated a DL method that can accurately predict whether the abnormal tissue is malignant or benign. Two deep 3D models along with “customised mixed link network (CMixNet)” designs were used for LN recognition and categorization. LN identification was performed using “Faster R-CNN” on efficiently learned features. LN Categorization was nodules was done by a Gradient Boosting Machine (GBM) on the learned features. It was validated using the open-source data set “Lung Image Database Consortium (LIDC)” which yielded a sensitivity of 94% and specificity of 91%.
To help CT reading process, Hongtao et al. [58] suggested a Two-dimensional CNN for LN detection. First, it modified the anatomy of “Faster R-CNN” LN detection by training three models for three different slice types. Second, a boosting architecture based on Two-dimensional CNN was created in order to reduce false positives from boosting results.
A deep lung nodule detection method has been developed by Xuechen et al. [59]. It extracted features using “Patch-based multi-resolution convolutional networks” and classified them by employing four distinct fusion approaches. The system performed substantially better and was much more robust than previous research. The method’s Free Response Area under Curve (FAUC) and “Refined Competition Performance Metrics” (R-CPM) were 0.982 and 0.987, respectively, and it also produced high False Nagative Rate (FNR) and FPR values, lowering efficiency.
Using a Three-Dimesnsional CNN with multi-scale prediction, Xiaoqi et al. [60] detected lung nodules. In addition, multi-scale LN prediction approach was also developed to detect extremely tiny nodules including “multi-scales cube prediction” and “cube clustering”. The sensitivity of the primary option system at one and four false positives is 87.94 percent and 92.93 percent, respectively. But Minute nodules could not be detected.
Huang et al. [61] created an end-to-end system that can segment exact pulmonary contours from raw thoracic CT scans in a quick and fully automated manner. The system’s four key components were LN identification with a faster regional-CNN (R-CNN), candidate melding, False Positive (FP) reduction with CNN, and LN segmentation with a tailored Fully Convolutional Neural Network (FCNN). The entire system is deviod of human involvement or database-specific design. On a standard workstation, each scan took about 16 seconds to complete. With a high average of false positives (FPs) per scan, nodule detection accuracy was 91.4 percent and 94.6 percent, respectively.
Peng Cao et al. [62] created a nodule classification method based on Deep CNN that had the benefit of auto learning representation and powerful generalisation ability. A systematic approach for identifying nodules of types solid, semisolid including Ground-Glass Opacity (GGO) have been developed. The LIDC database supplied 62,492 Regions-of-Interest (ROI)samples, including 40,772 nodules and 21,720 non nodules, which were utilised to train Deep CNN. Experiments showed that the method exhibited better sensitivity and accuracy, and outperformed other methods in race. But the method is heavily influenced by noise.
Cao et al. [63] offered a two-stage-CNN (TSCNN) for LN detection. For establishing an initial detection of LN, the enhanced U-Net network was formed. The next one was centred upon the developing dual pooling structure, which was constructed into 3 3-Dimensional CNN networks aimed at FP diminution. As the network training needed a significant quantity of training data, it devised an arbitrary mask as the data augmentation technique. Additionally, the generality ability of the FP diminution model was enhanced via ensemble learning.
Bobadilla et al. [64] presented a method to identify lung tumours that used CNNs. To compensate for the scarcity of nodule samples in comparison to background samples, training was performed that balanced the mini batches on each “Stochastic gradient descent (SGD)” iteration. The method outperformed a base feature-engineering method based on the same techniques for other stages LN detection, and CNNs outperformed the most recently developed methods on a dataset provided by the Japanese Society of Radiological Technology (JSRT).
The DL model designed by Zheng et al. [65] detects nodules in ultrasound images. The system was meant for both LN Detection and FP Reduction. To categorise axial, coronal, and sagittal views, an in-depth Encoder-Decoder network was trained. Then the possible LN from three planes were merged. To fine tune results, a Three-dimensional multiscale CNN is used to remove non-nodules. After the tenfold cross-validation of the LIDC-IDRI dataset, on 888 scans with 1186 nodules, the system was reviewed by at least three out of four radiologists. This led to problems because the training and testing process took a very long time to detect the anomaly. Table 5 illustrates Various LN detection techniques based on Deep CNN.
Table 5 Methods focussed on deep convolutional neural networks
| Reference | Technique | Advantages | Drawbacks |
| Nasrullah et al. [57] | CMixNet+R-CNN+GBM | false positive reduction at early stage and low time complexity | Cannot process reconstructed CT image at a time. |
| Hongtao et al. [58] | Detection framework based on 2D-CNN | It can accurately detect the latent pulmonary nodules | Accuracy can be improved further |
| Xuechen et al. [59] | Patch based multi resolution CNN | Higher Sensitivity and Low FPs/Image | Need to be tested on a real time large dataset. |
| Xiaoqi et al. [60] | 3D Deep CNNMulti scale Prediction | Nodule detection accuracy was promising | CNN considers only fewer parameters |
| Huang et al. [61] | R-CNN FCNN | Nodule detection accuracy was promising | Performance gap when compared with other methods |
| Peng Cao et al. [62] | Deep CNN with Auto learning capability | More Robust method | Limited to identify LN of types solid, semi solid and ground glass opacity |
| Cao et al. [63] | 2-Stage CNN with 3D CNN | Nodule detection accuracy was promising | Performance measures can be improved |
| Bobadilla et al. [64] | Deep CNN with SGD | Through data augmentation and dropout regularisation, CNN is effective LN identification | Only few features were extracted |
| Zheng et al. [65] | Deep CNN with multi planar nodule detection 3-D multiscale dense CNN | Capable to detect a greater number of nodules | may miss ground glass nodules. |
| Ling et al. [66] | 3D deep convolutional neural networks (CNNs) | Lesser parameters, faster convergence, and reduced over fitting problems | The stack of Inception layers resulted a reduction in network parameters and a high error rate. |
| Wang et al. [67] | deep convolutional neural networks (CNNs) | It vanished gradient issues and attained a low error rate | Misclassified the nodules and computational time was more |
| Mendoza et al. [68] | CNN | Accurate detection of nodule, error rate was reduced | Addicted towards various noise |
| Chen et al. [69] | Faster R-CNN | Nodule detection rate for various categories of nodule was high | High FRR and FPR values |
| Suresh et al. [70] | Deep-CNN | Accurate categorization of the nodule class without time complexity | Improper feature mapping |
| Ying et al. [71] | Faster R-CNN | Large nodules were detected easily | Poor reliability, highly complex |
4 Performance Comparison of Selected Works
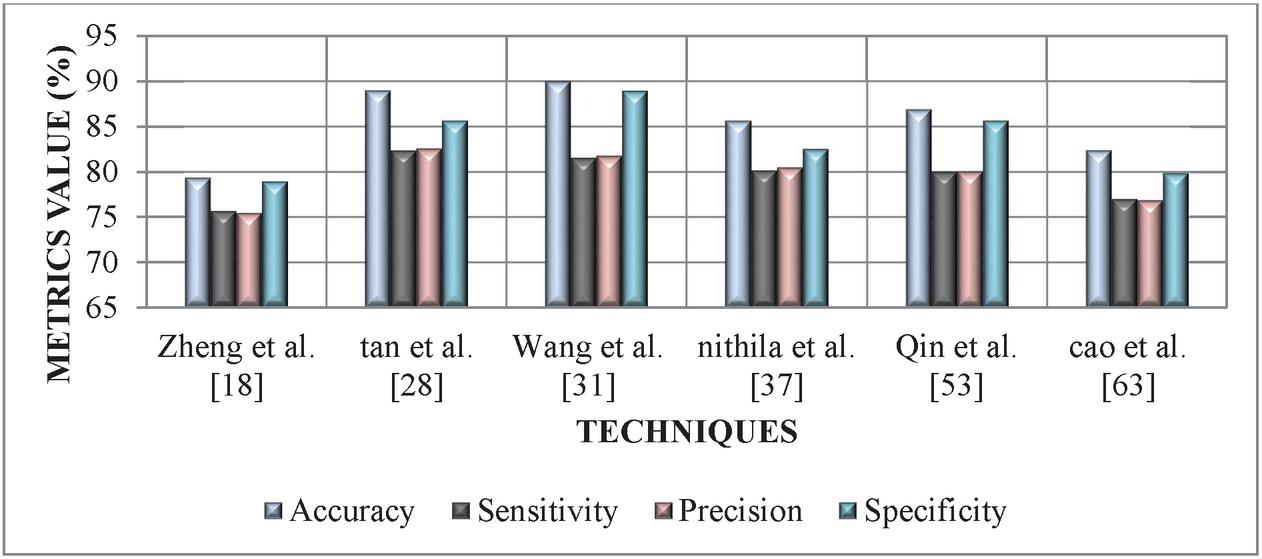
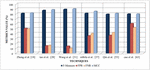
Various performance metrics, such as, FPR, Sensitivity, Accuracy Specificity, Precision, Matthew’s Correlation Coefficient (MCC) F-measure FNR of the selected works from the above-reviewed topics have been compared. The methods developed by the authors Zheng et al. [18], Tan et al. [28], Wang et al. [31], Nithila et al. [37], Qin et al. [53], and Cao et al. [63] tends to be more effective towards detecting the nodule and each technique is dependent on some stages, such as Segmentation, Feature Extraction, and Multistep Detection etc. The graphical representation of selected approaches based on the metrics is illustrated in the Figures 2 and 3.
Table 6 Performance metrics (accuracy, sensitivity, precision, specificity) of selected works
| Performance | Zheng | Tan | Wang | Nithila | Qin | Cao |
| Metrics/Techniques | et al. [18] | et al. [28] | et al. [31] | et al. [37] | et al. [53] | et al. [63] |
| Accuracy | 79.32 | 88.96 | 89.99 | 85.64 | 86.89 | 82.35 |
| Sensitivity | 75.65 | 82.35 | 81.52 | 80.12 | 79.99 | 76.96 |
| Precision | 75.45 | 82.56 | 81.75 | 80.45 | 80.01 | 76.85 |
| Specificity | 78.96 | 85.65 | 88.95 | 82.54 | 85.65 | 79.86 |
Figure 2 Performance comparison of various LN detection technique based on accuracy, specificity, sensitivity, and precision.
From graphical analysis in Figure 2 we can conclude that, Tan et al. [28] and Wang et al. [31], methods achieved better metrics values ranging between 81.52%–89.99% as compared to other methods for detecting lung nodules. Thereafter, Zheng et al. [18] and Cao et al. [63] approach tends to achieve the lowest metrics value ranging between 75.45%–82.35%. Finally, moderate performance is attained by the approach of Nithila et al. [37] and Qin et al. [53] with a metric value ranging between 79.99%–86.89%.
From the above comparison we can conclude that the automatic classification methods carried out in several stages (second group of methods) yielded better outcomes when complemented with other techniques.
Table 7 Performance metrics (F-Measure, FPR, FNR, MCC) of selected works
| Zheng | Tan | Wang | Nithila | Qin | Cao | |
| et al. [18] | et al. [28] | et al. [31] | et al. [37] | et al. [53] | et al. [63] | |
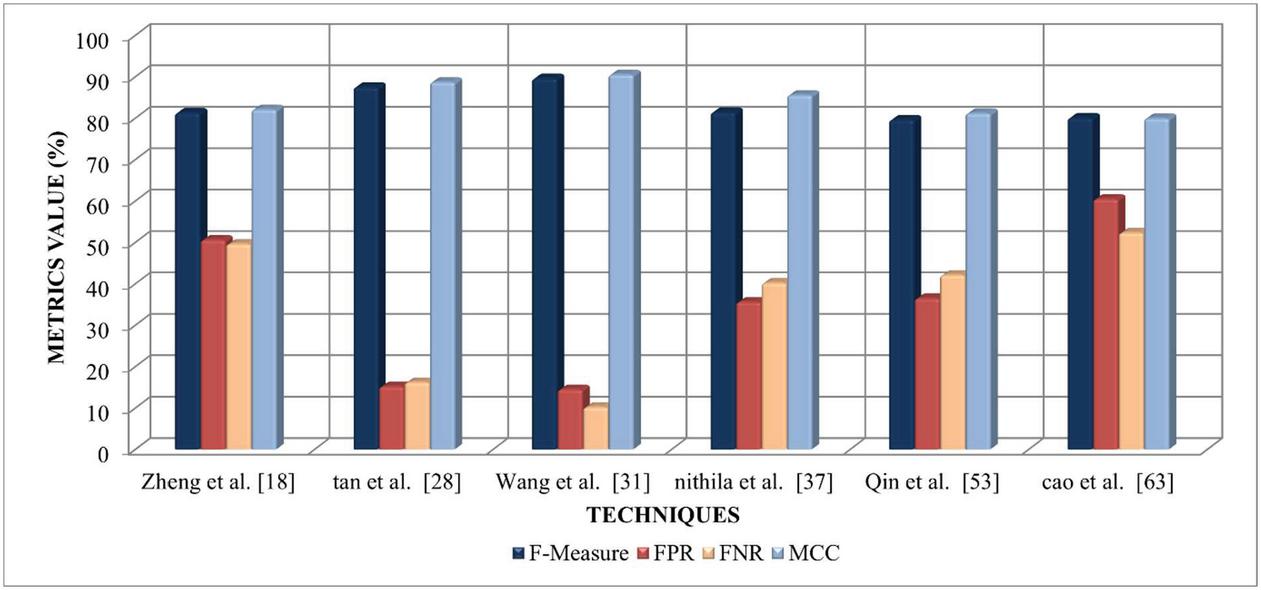
| F-Measure | 81.35 | 87.56 | 89.64 | 81.48 | 79.65 | 80.13 |
| FPR | 50.64 | 15.23 | 14.52 | 35.65 | 36.53 | 60.52 |
| FNR | 49.65 | 16.24 | 10.23 | 40.25 | 42.15 | 52.36 |
| MCC | 82.14 | 88.76 | 90.56 | 85.62 | 81.23 | 79.99 |
Figure 3 Performance comparison of selected LN detection technique based on F-measure, FPR, FNR, and MCC.
Figure 3 illustrates the performance comparison of selected LN detection techniques based on F-measure, FPR, FNR, and MCC. The graphical analysis infers that the Tan et al. [28] and Wang et al. [31], approaches achieved higher F-Measure and MCC value ranging between 87.56%–90.56% as compared to others. The methods developed by Zheng et al. [18] and Cao et al. [63] tends to achieve the highest FPR and FNR value ranging between 49.65% to 60.52%. Nithila et al. [37] and Qin et al. [53] approaches achieved an FPR and FNR value ranging between 35.65%–42.15%, which is better than Zheng et al. [18] and Cao et al. [63].
From the Performance Comparison of the selected works we can infer that all the methods are yet to achieve 100% accuracy. Performance metrics such as Specificity, Sensitivity and False positive reduction need to be improved further.
5 Conclusion
Detection of lung nodules has become essential for early diagnosis and medical management of lung cancer to avoid deaths. In this paper, recently developed deep learning methods were analysed and examined in detail, highlighting their advantages and drawbacks by dividing the approaches into five groups, namely, segmentation-based classification methods, feature extraction-based methods, multi-step automated detection methods, automated detection methods and deep convolutional neural-network based detection methods. It can be inferred that the automated multi-step processing of the nodule may be capable of obtaining a better detection rate of lung nodules. Although various methodologies have been developed, there is still a need for innovative methodologies to overcome the drawbacks highlighted in this paper. CNN’s have been shown to outperform other methods used for identifying lung nodules, so much of the research is focused on improving CNNs. An accuracy of 100% is yet to be achieved. To enhance the accuracy of lung cancer identification, additional patient details such as previous medical records and genetic profiles can be processed and blended with the intense features mined from CT images. Most of the approaches have been validated on datasets that are publicly available. These approaches need to be evaluated on real-time medical databases and this is a significant issue in medical research. Most researchers restricted their work to detecting lung nodules and their classification. it is possible to use DL techniques to automate lung cancer staging to get the severity levels of the cancer. Even though several investigators have used numerous DL models to increase the accuracy of lung nodule classification, there is still room for advancement in overcoming the above difficulties to identify malignant tumours at an early level.
References
[1] The National Lung Screening Trial Research Team. (2011). “Reduced lung-cancer mortality with low-dose computed tomographic screening”. New England Journal of Medicine, 365(5), 395–409.
[2] Rumelhart, D., Hinton, G. & Williams, R. “Learning representations by back-propagating errors”. Nature 323, 533–536 (1986). https://doi.org/10.1038/323533a0
[3] Fukushima, K. Neocognitron: A self-organizing neural network model for a mechanism of pattern recognition unaffected by shift in position. Biol. Cybernetics 36, 193–202 (1980). https://doi.org/10.1007/BF00344251.
[4] Hinton, G. E., & Salakhutdinov, R. R. (2006). “Reducing the dimensionality of data with neural networks”. Science, 313(5786), 504
[5] Goodfellow, Ian, Jean Pouget-Abadie, Mehdi Mirza, Bing Xu, David Warde-Farley, Sherjil Ozair, Aaron Courville, and Yoshua Bengio. (2014). “Generative Adversarial Nets”. Advances Neural Information Processing Systems, 2672–2680.
[6] Salakhutdinov, R., & Hinton, G. (2012). “An efficient learning procedure for deep Boltzmann machines”. Neural Computation, 24(8), 1967–2006.
[7] Sepp Hochreiter; Jürgen Schmidhuber (1997). “Long short-term memory”. Neural Computation. 9(8): 1735–1780. doi:10.1162/neco.1997.9.8. 1735.
[8] Broomhead, D.S., Lowe, David (1988). Radial basis functions, multi-variable functional interpolation and adaptive networks (Technical report).
[9] Broomhead, D.S., Lowe, David (1988). “Multivariable functional interpolation and adaptive networks”. Complex Systems. 2: 321–355.
[10] Schwenker F., Kestler H.A., Palm G. “Three learning phases for radial-basis-function networks.” Neural networks: the official journal of the International Neural Network Society, vol. 14,4–5 (2001): 439–58. doi:10.1016/s0893-6080(01)00027-2.
[11] Marius, Popescu & Balas, Valentina & Perescu-Popescu, Liliana & Mastorakis, Nikos. (2009). Multilayer perceptron and neural networks. WSEAS Transactions on Circuits and Systems. 8.
[12] Kohonen, T. Self-organized formation of topologically correct feature maps. Biol. Cybern. 43, 59–69 (1982). https://doi.org/10.1007/BF00337288.
[13] Tanzila Saba, “Automated lung nodule detection and classification based on multiple classifiers voting”, Microscopy Research and Technique, vol. 82, no. 9, pp. 1601–1609, 2019.
[14] Atsushi Teramoto and Hiroshi Fujita, “Automated lung nodule detection using positron emission tomography/computed tomography”, In Artificial Intelligence in Decision Support Systems For Diagnosis in Medical Imaging, Springer, Cham, pp. 87–110, 2018, 10.1007/978-3-319-68843-5_4.
[15] Shiwen Shen, Alex AT Bui, Jason Cong and William Hsu, “An automated lung segmentation approach using bidirectional chain codes to improve nodule detection accuracy”, Computers in Biology and Medicine, vol. 57, pp. 139–149, 2015, 10.1016/j.compbiomed.2014.12.008.
[16] Syed Muhammad Naqi, Muhammad Sharif and Ikram Ullah Lali, “A nodule candidate detection method supported by hybrid features to reduce false positives in lung nodule detection”, Multimedia Tools and Applications, vol. 78, no. 18, pp. 26287–26311, 2019.
[17] Qinghai Zhang and Xiaojing Kong, “Design of automatic lung nodule detection system based on multi-scene deep learning framework”, IEEE Access, vol. 8, pp. 90380–90389, 2020, 10.1109/ACCESS.2020.2993872.
[18] Sunyi Zheng, Jiapan Guo, Xiaonan Cui, Raymond NJ Veldhuis, Matthijs Oudkerk and Peter MA Van Ooijen, “Automatic pulmonary nodule detection in CT scans using convolutional neural networks based on maximum intensity projection”, IEEE Transactions on Medical Imaging, vol. 39, no. 3, pp. 797–805, 2019.
[19] Wenkai Huang and Lingkai Hu, “Using a noisy U-net for detecting lung nodule candidates”, IEEE Access, vol. 7, pp. 67905–67915, 2019, 10.1109/ACCESS.2019.2918224.
[20] Ling Fu, Jingchen Ma, Yacheng Ren, Youn Seon Han and Jun Zhao, “Automatic detection of lung nodules false positive reduction using convolution neural networks and handcrafted features”, In Medical Imaging Computer-Aided Diagnosis, International Society for Optics and Photonics, vol. 10134, pp. 101340A, 2017, 10.1117/12.2253995.
[21] Wei Shen, Mu Zhou, Feng Yang, Dongdong Yu, Di Dong, Caiyun Yang, Yali Zang and Jie Tian, “Multi-crop convolutional neural networks for lung nodule malignancy suspiciousness classification”, Pattern Recognition, vol. 61, pp. 663–673, 2017, 10.1016/j.patcog.2016.05.029.
[22] Hongyang Jiang, He Ma, Wei Qian, Mengdi Gao and Yan Li, “An automatic detection system of lung nodule based on multigroup patch-based deep learning network”, IEEE Journal of Biomedical and Health Informatics, vol. 22, no. 4, pp. 1227–1237, 2017.
[23] Wangxia Zuo, Fuqiang Zhou, Zuoxin Li and Lin Wang, “Multi-resolution CNN and knowledge transfer for candidate classification in lung nodule detection”, IEEE Access, vol. 7, pp. 32510–32521, 2019, 10.1109/ACCESS.2019.2903587.
[24] Tanzila Saba, Ahmed Sameh, Fatima Khan, Shafqat Ali Shad and Muhammad Sharif, “Lung nodule detection based on ensemble of hand crafted and deep features”, Journal of Medical Systems, vol. 43, no. 12, pp. 1–12, 2019.
[25] Talha Meraj, Hafiz Tayyab Rauf, Saliha Zahoor, Arslan Hassan, M. Ikram Ullah Lali, Liaqat Ali, Syed Ahmad Chan Bukhari and Umar Shoaib, “Lung nodules detection using semantic segmentation and classification with optimal features”, Neural Computing and Applications, pp. 1–14, 2019, 10.20944/preprints201909.0139.v1.
[26] Yuyun Ye, Miao Tian, Qiyu Liu and Heng-Ming Tai, “Pulmonary nodule detection using v-net and high-level descriptor based svm classifier”, IEEE Access, vol. 8, pp. 176033–176041, 2020, 10.1109/ACCESS.2020.3026168.
[27] Patrice Monkam, Shouliang Qi, Mingjie Xu, Haoming Li, Fangfang Han, Yueyang Teng and Wei Qian, “Ensemble learning of multiple-view 3D-CNNs model for micro-nodules identification in CT images”, IEEE Access, vol. 7, pp. 5564–5576, 2018, 10.1109/ACCESS.2018.2889350.
[28] Man Tan, Fa Wu, Bei Yang, Jinlian Ma, Dexing Kong, Zengsi Chen and Dan Long, “Pulmonary nodule detection using hybrid two-stage 3D CNNs”, Medical physics, vol. 47, no. 8, pp. 3376–3388, 2020.
[29] Zheng, Guangyuan, Guanghui Han, and Nouman Qadeer Soomro, “An inception module CNN classifiers fusion method on pulmonary nodule diagnosis by signs”, Tsinghua Science and Technology, vol. 25, no. 3, pp. 368–383, 2019.
[30] Lin Lu, Yongqiang Tan, Lawrence H. Schwartz and Binsheng Zhao, “Hybrid detection of lung nodules on CT scan images”, Medical Physics, vol. 42, no. 9, pp. 5042–5054, 2015.
[31] Xiuyuan Xu, Chengdi Wang, Jixiang Guo, Lan Yang, Hongli Bai, Weimin Li and Zhang Yi, “DeepLN a framework for automatic lung nodule detection using multi-resolution CT screening images”, Knowledge-Based Systems, vol. 189, pp. 105128, 2020, 10.1016/j.knosys.2019.105128.
[32] Imdad Ali, Muhammad Muzammil, Ihsan Ul Haq, Amir A. Khaliq and Suheel Abdullah, “Efficient lung nodule classification using transferable texture convolutional neural network”, IEEE Access, vol. 8, pp. 175859–175870, 2020, 10.1109/ACCESS.2020.3026080.
[33] Chung-Feng Jeffrey Kuo, Chang-Chiun Huang, Jing-Jhong Siao, Chia-Wen Hsieh, Vu Quang Huy, Kai-Hsiung Ko and Hsian-He Hsu, “Automatic lung nodule detection system using image processing techniques in computed tomography”, Biomedical Signal Processing and Control, vol. 56, pp. 101659, 2020, 10.1016/j.bspc.2019.101659.
[34] Syed Muhammad Naqi, Muhammad Sharif and Mussarat Yasmin, “Multistage segmentation model and SVM-ensemble for precise lung nodule detection”, International Journal of Computer Assisted Radiology and Surgery, vol. 13, no. 7, pp. 1083–1095, 2018.
[35] Yuanli Feng, Pengyi Hao, Peng Zhang, Xinguo Liu, Fuli Wu and Hongwei Wang, “Supervoxel based weakly-supervised multi-level 3D CNNs for lung nodule detection and segmentation”, Journal of Ambient Intelligence and Humanized Computing, pp. 1–11, 2019, 10.1007/s12652-018-01170-5.
[36] Jibi John and M. G. Mini, “Multilevel thresholding-based segmentation and feature extraction for pulmonary nodule detection”, Procedia Technology, vol. 24, pp. 957–963, 2016, 10.1016/j.protcy.2016.05.209.
[37] Ezhil E Nithila and Kumar S.S., “Segmentation of lung nodule in CT data using active contour model and Fuzzy C-mean clustering”, Alexandria Engineering Journal, vol. 55, no. 3, pp. 2583–2588, 2016.
[38] Amal Eisapour Moghaddam, Gholamreza Akbarizadeh and Hooman Kaabi, “Automatic detection and segmentation of blood vessels and pulmonary nodules based on a line tracking method and generalized linear regression model”, Signal, Image and Video Processing, vol. 13, no. 3, pp. 457–464, 2019.
[39] He Ren, Lingxiao Zhou, Gang Liu, Xueqing Peng, Weiya Shi, Huilin Xu, Fei Shan and Lei Liu, “An unsupervised semi-automated pulmonary nodule segmentation method based on enhanced region growing”, Quantitative Imaging in Medicine and Surgery, vol. 10, no. 1, pp. 233, 2020.
[40] Joana Rocha, António Cunha and Ana Maria Mendonça, “Conventional filtering versus u-net based models for pulmonary nodule segmentation in ct images”, Journal of Medical Systems, vol. 44, no. 4, pp. 1–8, 2020.
[41] Ji-kui Liu, Hong-yang Jiang, Chen-guang He, Yu Wang, Pu Wang and He Ma, “An assisted diagnosis system for detection of early pulmonary nodule in computed tomography images”, Journal of Medical Systems, vol. 41, no. 2, pp. 30, 2017.
[42] Benita K.J. Veronica, “An effective neural network model for lung nodule detection in CT images with optimal fuzzy model”, Multimedia Tools and Applications, pp. 1–21, 2020, 10.1007/s11042-020-08618-x.
[43] Haichao Cao, Hong Liu, Enmin Song, Chih-Cheng Hung, Guangzhi Ma, Xiangyang Xu, Renchao Jin and Jianguo Lu, “Dual-branch residual network for lung nodule segmentation”, Applied Soft Computing, vol. 86, pp. 105934, 2020,
[44] Erdal Taşcı and Aybars Uğur, “Shape and texture based novel features for automated juxtapleural nodule detection in lung CTs”, Journal of Medical Systems, vol. 39, no. 5, pp. 1–13, 2015,
[45] Furqan Shaukat, Gulistan Raja, Rehan Ashraf, Shehzad Khalid, Mudassar Ahmad and Amjad Ali, “Artificial neural network based classification of lung nodules in CT images using intensity shape and texture features”, Journal of Ambient Intelligence and Humanized Computing, vol. 10, no. 10, pp. 4135–4149, 2019.
[46] Syed Muhammad Naqi, Muhammad Sharif and Arfan Jaffar, “Lung nodule detection and classification based on geometric fit in parametric form and deep learning”, Neural Computing and Applications, vol. 32, no. 9, pp. 4629–4647, 2020.
[47] Changmiao Wang, Ahmed Elazab, Jianhuang Wu and Qingmao Hu, “Lung nodule classification using deep feature fusion in chest radiography”, Computerized Medical Imaging and Graphics, vol. 57, pp. 10–18, 2017, 10.1016/j.compmedimag.2016.11.004.
[48] Giovanni Lucca França da Silva, Thales Levi Azevedo Valente, Aristófanes Corrêa Silva, Anselmo Cardoso de Paiva and Marcelo Gattass, “Convolutional neural network-based PSO for lung nodule false positive reduction on CT images”, Computer Methods and Programs in Biomedicine, vol. 162, pp. 109–118, 2018, 10.1016/j.cmpb.2018.05.006.
[49] Furqan Shaukat, Gulistan Raja, Ali Gooya and Alejandro F. Frangi, “Fully automatic detection of lung nodules in CT images using a hybrid feature set”, Medical Physics, vol. 44, no. 7, pp. 3615–3629, 2017.
[50] Yutong Xie, Jianpeng Zhang, Yong Xia, Michael Fulham and Yanning Zhang, “Fusing texture shape and deep model-learned information at decision level for automated classification of lung nodules on chest CT”, Information Fusion, vol. 42, pp. 102–110, 2018, 10.1016/j.inffus.2017.10.005.
[51] Jingjing Yuan, Xinglong Liu, Fei Hou, Hong Qin and Aimin Hao, “Hybrid-feature-guided lung nodule type classification on CT images”, Computers & Graphics, vol. 70, pp. 288–299, 2018, 10.1016/j.cag.2017.07.020.
[52] Farahani F. V, Ahmadi A & Zarandi M. H. F, “Hybrid intelligent approach for diagnosis of the lung nodule from CT images using spatial kernelized fuzzy c-means and ensemble learning”, Mathematics and Computers in Simulation, vol. 149, pp. 48–68, 2018, 10.1016/j.matcom.2018.02.001.
[53] Yulei Qin, Hao Zheng, Xiaolin Huang, Jie Yang and Yue-Min Zhu, “Pulmonary nodule segmentation with CT sample synthesis using adversarial networks”, Medical Physics, vol. 46, no. 3, pp. 1218–1229, 2019.
[54] Dhara AK, Mukhopadhyay S, Dutta A, Garg M, Khandelwal N. A, “Combination of shape and texture features for classification of pulmonary nodules in lung CT images”, Journal of Digital Imaging, vol. 29, no. 4, pp. 466–75, 2016.
[55] Xuechen Li, Linlin Shen and Suhuai Luo, “A solitary feature-based lung nodule detection approach for chest X-ray radiographs”, IEEE journal of biomedical and health informatics, vol. 22, no. 2, pp. 516–524, 2017.
[56] Noor Khehrah, Muhammad Shahid Farid, Saira Bilal and Muhammad Hassan Khan, “Lung nodule detection in CT images using statistical and shape-based features”, Journal of Imaging, vol. 6, no. 2, pp. 6, 2020.
[57] Nasrullah N, Sang J , Alam M. S, Mateen M, Cai B & Hu H, “Automated lung nodule detection and classification using deep learning combined with multiple strategies”, Sensors, vol. 19, no. 17, pp. 3722, 2019.
[58] Hongtao Xie, Dongbao Yang, Nannan Sun, Zhineng Chen and Yongdong Zhang, “Automated pulmonary nodule detection in CT images using deep convolutional neural networks”, Pattern Recognition, vol. 85, pp. 109–119, 2019, 10.1016/j.patcog.2018.07.031.
[59] Xuechen Li, Linlin Shen, Xinpeng Xie, Shiyun Huang, Zhien Xie, Xian Hong and Juan Yu, “Multi-resolution convolutional networks for chest X-ray radiograph based lung nodule detection”, Artificial Intelligence in Medicine, vol. 103, pp. 101744, 2020, 10.1016/j.artmed.2019.101744.
[60] Yu Gu, Xiaoqi Lu, Lidong Yang, Baohua Zhang, Dahua Yu, Ying Zhao, Lixin Gao, Liang Wu and Tao Zhou, “Automatic lung nodule detection using a 3D deep convolutional neural network combined with a multi-scale prediction strategy in chest CTs”, Computers in Biology and Medicine, vol. 103, pp. 220–231, 2018, 10.1016/j.compbiomed.2018.10.011.
[61] Xia Huang, Wenqing Sun, Tzu-Liang Bill Tseng, Chunqiang Li and Wei Qian, “Fast and fully-automated detection and segmentation of pulmonary nodules in thoracic CT scans using deep convolutional neural networks”, Computerized Medical Imaging and Graphics, vol. 74, pp. 25–36, 2019, 10.1016/j.compmedimag.2019.02.003.
[62] Wei Li, Peng Cao, Dazhe Zhao and Junbo Wang, “Pulmonary nodule classification with deep convolutional neural networks on computed tomography images”, Computational and Mathematical Methods in Medicine, 2016, 10.1155/2016/6215085.
[63] Haichao Cao, Hong Liu, Enmin Song, Guangzhi Ma, Xiangyang Xu, Renchao Jin, Tengying Liu and Chih-Cheng Hung, “A two-stage convolutional neural networks for lung nodule detection”, IEEE Journal of Biomedical And Health Informatics, vol. 24, no. 7, pp. 2006–2015, 2020.
[64] Julio Cesar Mendoza Bobadilla and Helio Pedrini, “Lung nodule classification based on deep convolutional neural networks”, In Iberoamerican Congress on Pattern Recognition, Springer, Cham, pp. 117–124, 2016.
[65] Sunyi Zheng, Ludo J. Cornelissen, Xiaonan Cui, Xueping Jing, Raymond NJ Veldhuis, Matthijs Oudkerk and Peter MA van Ooijen, “Deep convolutional neural networks for multi-planar lung nodule detection improvement in small nodule identification”, Medical Physics, 2020, 10.1002/mp.14648.
[66] Ling Fu, Jingchen Ma, Yizhi Chen, Rasmus Larsson and Jun Zhao, “Automatic detection of lung nodules using 3d deep convolutional neural networks”, Journal of Shanghai Jiaotong University (Science), vol. 24, no. 4, pp. 517–523, 2019.
[67] Qin Wang, Fengyi Shen, Linyao Shen, Jia Huang and Weiguang Sheng, “Lung nodule detection in CT images using a raw patch-based convolutional neural network”, Journal of Digital Imaging, vol. 32, no. 6, pp. 971–979, 2019.
[68] Julio Mendoza and Helio Pedrini, “Detection and classification of lung nodules in chest X-ray images using deep convolutional neural networks”, Computational Intelligence, vol. 36, no. 2, pp. 370–401, 2020.
[69] Sheng Chen, Yaqi Han, Jinqiu Lin, Xiangyu Zhao and Ping Kong, “Pulmonary nodule detection on chest radiographs using balanced convolutional neural network and classic candidate detection”, Artificial Intelligence in Medicine, vol. 107, pp. 10188, 2020, 10.1016/j.artmed.2020.101881.
[70] Supriya Suresh and Subaji Mohan, “NROI based feature learning for automated tumor stage classification of pulmonary lung nodules using deep convolutional neural networks”, Journal of King Saud University-Computer and Information Sciences, 2019, 10.1016/j.jksuci.2019.11.013.
[71] Ying Su, Dan Li and Xiaodong Chen, “Lung nodule detection based on faster r-cnn framework”, Computer Methods and Programs in Biomedicine, pp. 105866, 2020, 10.1016/j.cmpb.2020.105866.
Biographies

Pavan Kumar Illa Completed his master’s degree in Computer Science & Engineering. From K L University. He is Currently associated with Department of Computer Science & Engineering, SRMIST, Chennai as a Doctoral student and working as Assistant Professor in VNRVJIET, Hyderabad. His research interests include Machine Learning and Deep learning. He has 9 years of teaching & research experience.

T. Senthil Kumar Completed Ph.D. in the field of wireless communication, under the guidance of Dr.S. Prabhakaran, Professor, Department of Computer Science & Engineering, SRMIST, Chennai. He is currently working as Assistant Professor (Senior Grade) in SRMIST, Chennai. He has contributed many scientific research papers. His research interests include wireless communication, Machine Learning and Deep learning.

F. Syed Anwar Hussainy, Completed his master’s in information technology. He is Currently associated with SRMIST, Chennai as a Doctoral student. His research interests include Machine Learning and Deep learning
Journal of Mobile Multimedia, Vol. 18_2, 421–450.
doi: 10.13052/jmm1550-4646.18213
© 2021 River Publishers