Mechanisms, Models, and Clinical Applications of Cell Membrane Electroporation
DOI:
https://doi.org/10.13052/ijts2246-8765.2024.041Keywords:
Electroporation, nuclear and cell membrane permeability, drug delivery, mathematical modeling, cancer therapyAbstract
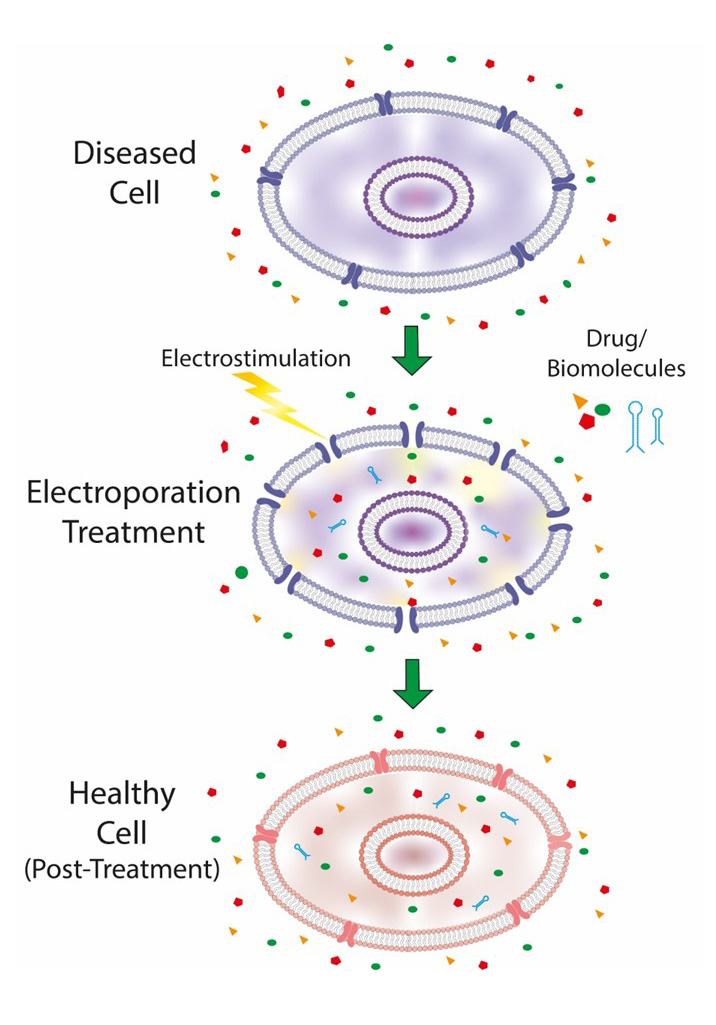
Electroporation is an essential biophysical process that involves the use of pulsed electric fields to temporarily increase the permeability of cell membranes. A comprehensive overview of the main clinical and biomedical applications of cellular electroporation is provided here with a particular focus on cancer therapy, genetics, and drug delivery. The review concentrates on the characterization of membrane stresses caused by electroporation and their impact on cell membrane structure and dynamics. It analyses the relationship between applied electric fields, cell geometry, and membrane composition with the aim of developing mathematical models to simulate cell geometries and the electroporation process. Furthermore, it identifies both the beneficial effects and potential complications of the electroporation treatment, as well as the various mathematical models that have been developed to simulate the effects of such treatments. The use of sophisticated simulation algorithms enables an in-depth investigation of the intricate relationship between electrical parameters and cellular responses, facilitating a comprehensive assessment of the efficacy and safety of the electroporation procedures.
Downloads
References
S. J. Singer e G. L. Nicolson, «The Fluid Mosaic Model of the Structure of Cell Membranes», Science, vol. 175, fasc. 4023, pp. 720–731, feb. 1972, doi: 10.1126/science.175.4023.720.
K. Simons e E. Ikonen, «Functional rafts in cell membranes», Nature, vol. 387, fasc. 6633, pp. 569–572, giu. 1997, doi: 10.1038/42408.
Lingwood, D., and Simons, K. (2010). Lipid rafts as a membrane-organizing science, 327(5961), 46–50.
W. A. Catterall, «From Ionic Currents to Molecular Mechanisms: The Structure and Function of Voltage-Gated Sodium Channels», Neuron, vol. 26, fasc. 1, pp. 13–25, apr. 2000, doi: 10.1016/S0896-6273(00)81133-2.
Staub, O., and Rotin, D. (2006). Role of ubiquitylation in cellular membrane transport. Physiological reviews, 86(2), 669–707.
W. A. Catterall, «Structure and Regulation of Voltage-Gated Ca2+ Channels», Annual Review of Cell and Developmental Biology, vol. 16, fasc. Volume 16, 2000, pp. 521–555, nov. 2000, doi: 10.1146/annurev.cellbio.16.1.521.
S. Tan, T. Wu, D. Zhang, e Z. Zhang, «Cell or Cell Membrane-Based Drug Delivery Systems», Theranostics, vol. 5, fasc. 8, p. 863, 2015, doi: 10.7150/thno.11852.
T. Batista Napotnik, T. Polajžer, e D. Miklavèiè, «Cell death due to electroporation – A review», Bioelectrochemistry, vol. 141, p. 107871, ott. 2021, doi: 10.1016/j.bioelechem.2021.107871.
T. Kotnik, P. Kramar, G. Pucihar, D. Miklavcic, e M. Tarek, «Cell membrane electroporation- Part 1: The phenomenon», IEEE Electrical Insulation Magazine, vol. 28, fasc. 5, pp. 14–23, set. 2012, doi: 10.1109/MEI.2012.6268438.
M. A. Chiapperino, P. Bia, C. M. Lamacchia, e L. Mescia, «Electroporation Modelling of Irregular Nucleated Cells Including Pore Radius Dynamics», Electronics, vol. 8, fasc. 12, Art. fasc. 12, dic. 2019, doi: 10.3390/electronics8121477.
C. Zhou, Z. Yan, e K. Liu, «Response characteristics and optimization of electroporation: simulation based on finite element method», Electromagnetic Biology and Medicine, vol. 40, fasc. 3, pp. 321–337, lug. 2021, doi: 10.1080/15368378.2021.1951484.
R. P. Joshi e K. H. Schoenbach, «Electroporation dynamics in biological cells subjected to ultrafast electrical pulses: A numerical simulation study», Phys. Rev. E, vol. 62, fasc. 1, pp. 1025–1033, lug. 2000, doi: 10.1103/PhysRevE.62.1025.
E. Langhoff e J. Ladefoged, «Sodium activity, sodium concentration, and osmolality in plasma in acute and chronic renal failure», Clin Chem, vol. 31, fasc. 11, pp. 1811–1814, nov. 1985.
M. Zacchia, M. L. Abategiovanni, S. Stratigis, e G. Capasso, «Potassium: From Physiology to Clinical Implications», Kidney Diseases, vol. 2, fasc. 2, p. 72, mag. 2016, doi: 10.1159/000446268.
T. Kotnik, L. Rems, M. Tarek, e D. Miklavèiè, «Membrane Electroporation and Electropermeabilization: Mechanisms and Models», Annual Review of Biophysics, vol. 48, fasc. Volume 48, 2019, pp. 63–91, mag. 2019, doi: 10.1146/annurev-biophys-052118-115451.
Goldberg, E., Suárez, C., Alfonso, M., Marchese, J., Soba, A., and Marshall, G. (2018). Cell membrane electroporation modeling: A multiphysics Bioelectrochemistry, 124, 28–39.
Napotnik, T. B., and Miklavèiè, D. (2018). In vitro electroporation detection methods – An overview. Bioelectrochemistry, 120, 166–182.
Liu, J., Jiang, J., He, M., Chen, J., Huang, S., Liu, Z., … and Wang, J. (2023). Nanopore Electroporation Device for DNA Transfection into Various Spreading and Nonadherent Cell Types. ACS Applied Materials & Interfaces, 15(43), 50015–50033.
Zeller, R. W. (2018). Electroporation in ascidians: history, theory and Transgenic ascidians, 37–48.
G. Pucihar, T. Kotnik, e D. Miklavèiè, «Measuring the Induced Membrane Voltage with Di-8-ANEPPS», JoVE (Journal of Visualized Experiments), fasc. 33, p. e1659, nov. 2009, doi: 10.3791/1659.
Gehl, J. J. A. P. S. (2003). Electroporation: theory and methods, perspectives for drug delivery, gene therapy and research. Acta Physiologica Scandinavica, 177(4), 437–447.
S. Skorupska, I. Grabowska-Jadach, A. Dybko, e Z. Brzozka, «Studies on electroporation and electrochemotherapy of adherent cells monolayer using electrode modules of specific geometry», Sensors and Actuators B: Chemical, vol. 351, p. 130889, gen. 2022, doi: 10.1016/j.snb.2021.130889.
Evangelopoulos, M., Yazdi, I. K., Acciardo, S., Palomba, R., Giordano, F., Pasto, A., … and Tasciotti, E. (2020). Biomimetic cellular vectors for enhancing drug delivery to the lungs. Scientific Reports, 10(1), 172.
McBride, J. L., Boudreau, R. L., Harper, S. Q., Staber, P. D., Monteys, A. M., Martins, I., … and Davidson, B. L. (2008). Artificial miRNAs mitigate shRNA-mediated toxicity in the brain: implications for the therapeutic development of Proceedings of the National Academy of 105(15), 5868–5873.
Neumann, E., Schaefer−
Ridder, M., Wang, Y., and Hofschneider, P. (1982). Gene transfer into mouse lyoma cells by electroporation in high electric fields. The EMBO journal, 1(7), 841–845.
M. Breton e L. M. Mir, «Microsecond and nanosecond electric pulses in cancer treatments», Bioelectromagnetics, vol. 33, fasc. 2, pp. 106–123, 2012, doi: 10.1002/bem.20692.
Sersa, G., Kranjc, S., Scancar, J., Krzan, M., and Cemazar, M. (2010). Electrochemotherapy of mouse sarcoma tumors using electric pulse trains with repetition frequencies of 1 Hz and 5 kHz. The Journal of membrane biology, 236, 155–162.
Zhang, Z., Huang, G., Li, Y., Chen, X., Yao, Y., Ren, S., … and An, C. (2022). Electrically conductive inorganic membranes: A review on principles, characteristics and applications. Chemical Engineering Journal, 427, 131987.
F. Liu, R. Su, X. Jiang, S. Wang, W. Mu, e L. Chang, «Advanced micro/nano-electroporation for gene therapy: recent advances and future outlook», Nanoscale, vol. 16, fasc. 22, pp. 10500–10521, giu. 2024, doi: 10.1039/D4NR01408A.
K. Balantiè, D. Miklavèiè, I. Križaj, e P. Kramar, «The Good and the Bad of Cell Membrane Electroporation», ACSi, vol. 68, fasc. 4, pp. 753–764, dic. 2021, doi: 10.17344/acsi.2021.7198.
Spugnini, E. P., Condello, M., Crispi, S., and Baldi, A. (2024). Electroporation in Translational Medicine: From Veterinary Experience to Human Oncology. Cancers, Cancers, 16(5), 1067.
Bo, W., Silkunas, M., Mangalanathan, U., Novickij, V., Casciola, M., Semenov, I., … and Pakhomov, A. G. (2020). Probing Nanoelectroporation and Resealing of the Cell Membrane by the Entry of Ca2+
and Ba2+
Ions. International Journal of Molecular Sciences, 21(9), 3386.
Vorobiev, E., Lebovka, N., Vorobiev, E., and Lebovka, N. (2020). Fundamentals of electroporation, theory and mathematical models for simulation of PEE processing. Processing of Foods and Biomass Feedstocks by Pulsed Electric Energy, 27–49.
S. Chopra, P. Ruzgys, M. Maciulevièius, M. Jakutavièiûtë, e S. Ðatkauskas, «Investigation of Plasmid DNA Delivery and Cell Viability Dynamics for Optimal Cell Electrotransfection In Vitro», Applied Sciences, vol. 10, fasc. 17, Art. fasc. 17, gen. 2020, doi: 10.3390/app10176070.
Y. Cu et al., «Enhanced Delivery and Potency of Self-Amplifying mRNA Vaccines by Electroporation in Situ», Vaccines (Basel), vol. 1, fasc. 3, pp. 367–383, ago. 2013, doi: 10.3390/vaccines1030367.
R. S. Son, K. C. Smith, T. R. Gowrishankar, P. T. Vernier, e J. C. Weaver, «Basic Features of a Cell Electroporation Model: Illustrative Behavior for Two Very Different Pulses», J Membrane Biol, vol. 247, fasc. 12, pp. 1209–1228, dic. 2014, doi: 10.1007/s00232-014-9699-z.
Chen, Y., Moser, M. A., Luo, Y., Zhang, W., and Zhang, B. (2019). Chemical enhancement of irreversible electroporation: A review and future suggestions. Technology in Cancer Research & Treatment, 18, 1533033819874128.
Chang, A. Y., Liu, X., Tian, H., Hua, L., Yang, Z., and Wang, S. (2020). Microfluidic electroporation coupling pulses of nanoseconds and milliseconds to facilitate rapid uptake and enhanced expression of DNA in cell therapy. Scientific reports, 10(1), 6061.
L. Lambricht, A. Lopes, S. Kos, G. Sersa, V. Préat, e G. Vandermeulen, «Clinical potential of electroporation for gene therapy and DNA vaccine delivery», Expert Opinion on Drug Delivery, vol. 13, fasc. 2, pp. 295–310, feb. 2016, doi: 10.1517/17425247.2016.1121990.
K. E. Broderick e L. M. Humeau, «Electroporation-enhanced delivery of nucleic acid vaccines», Expert Review of Vaccines, vol. 14, fasc. 2, pp. 195–204, feb. 2015, doi: 10.1586/14760584.2015.990890.
J. Jiang et al., «Integration of needle-free jet injection with advanced electroporation delivery enhances the magnitude, kinetics, and persistence of engineered DNA vaccine induced immune responses», Vaccine, vol. 37, fasc. 29, pp. 3832–3839, giu. 2019, doi: 10.1016/j.vaccine.2019.05.054.
K. R. Porter e K. Raviprakash, «DNA Vaccine Delivery and Improved Immunogenicity», Current Issues in Molecular Biology, vol. 22, fasc. 1, Art. fasc. 1, apr. 2017, doi: 10.21775/cimb.022.129.
Ledesma-Feliciano, C., Chapman, R., Hooper, J. W., Elma, K., Zehrung, D., Brennan, M. B., and Spiegel, E. K. (2023). Improved DNA vaccine delivery with needle-free injection systems. Vaccines, 11(2), 280.
Lu, B., Lim, J. M., Yu, B., Song, S., Neeli, P., Sobhani, N., … and Chai, D. (2024). The next-generation DNA vaccine platforms and delivery systems: Advances, challenges and prospects. Frontiers in immunology, 15, 1332939.
J. B. Sandbrink e G. D. Koblentz, «Biosecurity risks associated with vaccine platform technologies», Vaccine, vol. 40, fasc. 17, pp. 2514–2523, apr. 2022, doi: 10.1016/j.vaccine.2021.02.023.
D. N. Kisakov, I. M. Belyakov, L. A. Kisakova, V. A. Yakovlev, E. V. Tigeeva, e L. I. Karpenko, «The use of electroporation to deliver DNA-based vaccines», Expert Review of Vaccines, vol. 23, fasc. 1, pp. 102–123, dic. 2024, doi: 10.1080/14760584.2023.2292772.
A. Cvetkoska, E. Pírc, M. Reberšek, R. Magjarević, e D. Míklavèiè, «Towards standardization of electroporation devices and protocols», IEEE Instrumentation & Measurement Magazine, vol. 23, fasc. 2, pp. 74–81, apr. 2020, doi: 10.1109/MIM.2020.9062692.
Pisani, S., Bertino, G., Prina-Mello, A., Locati, L. D., Mauramati, S., Genta, I., … and Benazzo, M. (2022). Electroporation in head-and-neck cancer: an innovative approach with immunotherapy and nanotechnology combination. Cancers, Cancers, 14(21), 5363.
Rakoczy, K., Kisielewska, M., Sêdzik, M., Jonderko, L., Celińska, J., Sauer, N., … and Kulbacka, J. (2022). Electroporation in clinical applications – The potential of gene electrotransfer and electrochemotherapy. Applied Sciences, 12(21), 10821.
R. Upasani, A. Herwadkar, N. Singh, e A. K. Banga, «Innovations and Future Prospects of Dermal Delivery Systems», in Dermal Drug Delivery, CRC Press, 2020.
M. Liu et al., «Recent advances and research progress on microsystems and bioeffects of terahertz neuromodulation», Microsyst Nanoeng, vol. 9, fasc. 1, pp. 1–17, nov. 2023, doi: 10.1038/s41378-023-00612-1.
F. Saade e N. Petrovsky, «Technologies for enhanced efficacy of DNA vaccines», Expert Review of Vaccines, vol. 11, fasc. 2, pp. 189–209, feb. 2012, doi: 10.1586/erv.11.188.
Amanat, F., and Krammer, F. (2020). SARS-CoV-2 vaccines: status Immunity, 52(4), 583–589.
Z. Xu et al., «Harnessing Recent Advances in Synthetic DNA and Electroporation Technologies for Rapid Vaccine Development Against COVID-19 and Other Emerging Infectious Diseases», Front. Med. Technol., vol. 2, ott. 2020, doi: 10.3389/fmedt.2020.571030.
F. D’Alessio et al., «Immunogenicity of COVID-eVax Delivered by Electroporation Is Moderately Impacted by Temperature and Molecular Isoforms», Vaccines, vol. 11, fasc. 3, Art. fasc. 3, mar. 2023, doi: 10.3390/vaccines11030678.
L. Aurisicchio et al., A first-in-human trial on the safety and immunogenicity of COVID-eVax, a cellular response-skewed DNA vaccine against COVID-19, Mol Ther, vol. 31, fasc. 3, pp. 788–800, mar. 2023, doi: 10.1016/j.ymthe.2022.12.017.
A Mathematical Model to Expedite Electroporation Based Vaccine Development for COVID-19, Biointerface Res Appl Chem, vol. 12, fasc. 2, pp. 1951–1961, giu. 2021, doi: 10.33263/BRIAC122.19511961.
C. He, X. Huang, Y. Zhang, X. Lin, e S. Li, T-cell activation and immune memory enhancement induced by irreversible electroporation in pancreatic cancer, Clinical and Translational Medicine, vol. 10, fasc. 2, p. e39, 2020, doi: 10.1002/ctm2.39.
A. Gajewska-Naryniecka, U. Szwedowicz, Z. Łapińska, J. Rudno-Rudzińska, W. Kielan, e J. Kulbacka, Irreversible Electroporation in Pancreatic Cancer – An Evolving Experimental and Clinical Method, International Journal of Molecular Sciences, vol. 24, fasc. 5, Art. fasc. 5, gen. 2023, doi: 10.3390/ijms24054381.
H. Wang et al., «Extended Focal Ablation of Localized Prostate Cancer With High-Frequency Irreversible Electroporation: A Nonrandomized Controlled Trial», JAMA Surgery, vol. 157, fasc. 8, pp. 693–700, ago. 2022, doi: 10.1001/jamasurg.2022.2230.
P. Charalambous et al., «The efficacy and safety of the open approach irreversible electroporation in the treatment of pancreatic cancer: A systematic review», European Journal of Surgical Oncology, 46, fasc. 9, pp. 1565–1572, set. 2020, doi: 10.1016/j.ejso.2020.05.017.
Z. L. Rai, R. Feakins, L. J. Pallett, D. Manas, e B. R. Davidson, Irreversible Electroporation (IRE) in Locally Advanced Pancreatic Cancer: A Review of Current Clinical Outcomes, Mechanism of Action and Opportunities for Synergistic Therapy, Journal of Clinical Medicine, vol. 10, fasc. 8, Art. fasc. 8, gen. 2021, doi: 10.3390/jcm10081609.
A. Gyftopoulos, I. A. Ziogas, A. S. Barbas, e D. Moris, The Synergistic Role of Irreversible Electroporation and Chemotherapy for Locally Advanced Pancreatic Cancer, Front. Oncol., vol. 12, mag. 2022, doi: 10.3389/fonc.2022.843769.
A. Kiełbik, W. Szlasa, J. Saczko, e J. Kulbacka, Electroporation-Based Treatments in Urology, Cancers, vol. 12, fasc. 8, Art. fasc. 8, ago. 2020, doi: 10.3390/cancers12082208.
L. Lunelli, O. Cussenot, e J. J. M. C. H. de la Rosette, «Irreversible Electroporation (IRE) for Prostate Cancer», in Interventional Urology, A. R. Rastinehad, D. N. Siegel, B. J. Wood, e T. McClure, A c. di, Cham: Springer International Publishing, 2021, pp. 241–247. doi: 10.1007/978-3-030-73565-4_16.
Yu, M., and Li, S. (2022). Irreversible electroporation for liver cancer ablation: A meta analysis. European Journal of Surgical Oncology, 48(6), 1321–1330.
Z.-G. Liu, X.-H. Chen, Z.-J. Yu, J. Lv, e Z.-G. Ren, «Recent progress in pulsed electric field ablation for liver cancer», World J Gastroenterol, vol. 26, fasc. 24, pp. 3421–3431, giu. 2020, doi: 10.3748/wjg.v26.i24.3421.
D. Cano, J. J. Lasarte, e I. Vivas, [Irreversible electroporation: present and future in the treatment of hepatocellular carcinoma, An Sist Sanit Navar, vol. 45, fasc. 3, p. e1019, nov. 2022, doi: 10.23938/assn.1019.
J. Yang, A. Eresen, J. Shangguan, Q. Ma, V. Yaghmai, e Z. Zhang, Irreversible electroporation ablation overcomes tumor-associated immunosuppression to improve the efficacy of DC vaccination in a mice model of pancreatic cancer, OncoImmunology, vol. 10, fasc. 1, p. 1875638, gen. 2021, doi: 10.1080/2162402X.2021.1875638.
A. Nikyar e A. Bolhassani, Electroporation: An Effective Method For In Vivo Gene Delivery, Drug Delivery Letters, vol. 12, fasc. 1, pp. 35–45, mar. 2022, doi: 10.2174/2210303112666220127113328.
Go, E. J., Yang, H., Chon, H. J., Yang, D., Ryu, W., Kim, D. H., … and Park, W. (2020). Combination of irreversible electroporation and STING agonist for effective cancer immunotherapy. Cancers, 12(11), 3123.
M. B. Bendix, A. Houston, P. F. Forde, e E. Brint, Defining optimal parameters to maximize the effect of electrochemotherapy on lung cancer cells whilst preserving the integrity of immune cells, Bioelectrochemistry, vol. 148, p. 108257, dic. 2022, doi: 10.1016/j.bioelechem.2022.108257.
Vitkin, E., Singh, A., Wise, J., Ben-Elazar, S., Yakhini, Z., and Golberg, A. (2022). Nondestructive protein sampling with electroporation facilitates profiling of spatial differential protein expression in breast tumors in vivo. Scientific Reports, 12(1), 15835.
Z. Łapińska, U. Szwedowicz, A. Choromańska, e J. Saczko, Electroporation and Electrochemotherapy in Gynecological and Breast Cancer Treatment, Molecules, vol. 27, fasc. 8, Art. fasc. 8, gen. 2022, doi: 10.3390/molecules27082476.
N. A. Abd Rahman, M. M. Abdul Jamil, M. N. Adon, C. Chang Choon, e R. Ambar, Electroporation Study: Pulse Electric Field Effect on Breast Cancer Cell, in Proceedings of the 11th National Technical Seminar on Unmanned System Technology 2019, Z. Md Zain, H. Ahmad, D. Pebrianti, M. Mustafa, N. R. H. Abdullah, R. Samad, e M. Mat Noh, A c. di, Singapore: Springer Nature, 2021, pp. 751–760. doi: 10.1007/978-981-15-5281-6_54.
Ferioli, M., Perrone, A. M., Buwenge, M., Arcelli, A., Zamagni, A., Macchia, G., … and Morganti, A. G. (2021). Electrochemotherapy of skin metastases from breast cancer: a systematic review. Clinical & Experimental Metastasis, 38, 1–10.
R. V. Davalos, B. Rubinsky, e L. M. Mir, Theoretical analysis of the thermal effects during in vivo tissue electroporation, Bioelectrochemistry, vol. 61, fasc. 1, pp. 99–107, ott. 2003, doi: 10.1016/j.bioelechem.2003.07.001.
Davalos, R. V., Bhonsle, S., and Neal, R. E. (2015). Implications and considerations of thermal effects when applying irreversible electroporation tissue ablation The Prostate, 75(10), 1114–1118.
N. Beitel-White et al., Multi-Tissue Analysis on the Impact of Electroporation on Electrical and Thermal Properties, IEEE Transactions on Biomedical Engineering, vol. 68, fasc. 3, pp. 771–782, mar. 2021, doi: 10.1109/TBME.2020.3013572.
E. van Veldhuisen, J. A. Vogel, J. H. Klaessens, e R. M. Verdaasdonk, Thermal Effects of Irreversible Electroporation, in Irreversible Electroporation in Clinical Practice, M. R. Meijerink, H. J. Scheffer, e G. Narayanan, A c. di, Cham: Springer International Publishing, 2018, pp. 121–136. doi: 10.1007/978-3-319-55113-5_9.
K. Kurata, S. Nomura, e H. Takamatsu, «Three-dimensional analysis of irreversible electroporation: Estimation of thermal and non-thermal damage», International Journal of Heat and Mass Transfer, vol. 72, pp. 66–74, mag. 2014, doi: 10.1016/j.ijheatmasstransfer.2013.12.065.
Mahniè-Kalamiza, S., Ulrih, N. P., Vorobiev, E., and Miklavèiè, D. (2017). A comprehensive theoretical study of thermal relations in plant tissue following electroporation. International Journal of Heat and Mass Transfer, 111, 150–162.
P. Agnass et al., Mathematical modeling of the thermal effects of irreversible electroporation for in vitro, in vivo, and clinical use: a systematic review, International Journal of Hyperthermia, vol. 37, fasc. 1, pp. 486–505, gen. 2020, doi: 10.1080/02656736.2020.1753828.
A. Golberg e M. L. Yarmush, Nonthermal Irreversible Electroporation: Fundamentals, Applications, and Challenges, IEEE Transactions on Biomedical Engineering, 60, fasc. 3, pp. 707–714, mar. 2013, doi: 10.1109/TBME.2013.2238672.
Potter, H., and Heller, R. (2018). Transfection by electroporation. Current protocols in molecular biology, 121(1), 9–3.
S. N. Campelo, I. V. Edward J Jacobs, K. N. Aycock, e R. V. Davalos, Real-Time Temperature Rise Estimation during Irreversible Electroporation Treatment through State-Space Modeling, Bioengineering, vol. 9, fasc. 10, p. 499, set. 2022, doi: 10.3390/bioengineering9100499.
P. Agnass et al., HyCHEED System for Maintaining Stable Temperature Control during Preclinical Irreversible Electroporation Experiments at Clinically Relevant Temperature and Pulse Settings, Sensors (Basel, Switzerland), vol. 20, fasc. 21, p. 6227, ott. 2020, doi: 10.3390/s20216227.
B. Geboers et al., High-Voltage Electrical Pulses in Oncology: Irreversible Electroporation, Electrochemotherapy, Gene Electrotransfer, Electrofusion, and Electroimmunotherapy, Radiology, vol. 295, fasc. 2, pp. 254–272, mag. 2020, doi: 10.1148/radiol.2020192190.
Lyons, P., Polini, D., Russell-Ryan, K., and Clover, A. J. P. (2023). High-frequency electroporation and chemotherapy for the treatment of cutaneous malignancies: evaluation of early clinical response. Cancers, 15(12), 3212.
W. Lou et al., Present and future of metal nanoparticles in tumor ablation therapy, Nanoscale, vol. 15, fasc. 44, pp. 17698–17726, nov. 2023, doi: 10.1039/D3NR04362B.
Kwak, K., Yu, B., Lewandowski, R. J., and Kim, D. H. (2022). Recent progress in cryoablation cancer therapy and nanoparticles mediated Theranostics, 12(5), 2175.
C. C. Fesmire, R. H. Williamson, R. A. Petrella, J. D. Kaufman, N. Topasna, e M. B. Sano, Integrated Time Nanosecond Pulse Irreversible Electroporation (INSPIRE): Assessment of Dose, Temperature, and Voltage on Experimental and Clinical Treatment Outcomes, IEEE Transactions on Biomedical Engineering, vol. 71, fasc. 5, pp. 1511–1520, mag. 2024, doi: 10.1109/TBME.2023.3340718.
V. Novickij, N. Rembiałkowska, W. Szlasa, e J. Kulbacka, Does the shape of the electric pulse matter in electroporation?, Front. Oncol., vol. 12, set. 2022, doi: 10.3389/fonc.2022.958128.
Fait, T., Baltazár, T., Bubenickova, L., Kestranek, J., Stepan, M., Muller, M., and Turcan, P. (2023). Treatment of Vulvovaginal Laxity by Electroporation: The Jett Plasma Medical for Her II Study. Journal of Clinical Medicine, 12(19), 6234.
S. Etienne-Manneville, Microtubules in Cell Migration, Annual Review of Cell and Developmental Biology, vol. 29, fasc. Volume 29, 2013, pp. 471–499, ott. 2013, doi: 10.1146/annurev-cellbio-101011-155711.
R. Baskar, J. Dai, N. Wenlong, R. Yeo, e K.-W. Yeoh, Biological response of cancer cells to radiation treatment, Front. Mol. Biosci., vol. 1, nov. 2014, doi: 10.3389/fmolb.2014.00024.
J. Guck et al., Optical Deformability as an Inherent Cell Marker for Testing Malignant Transformation and Metastatic Competence, Biophysical Journal, vol. 88, fasc. 5, pp. 3689–3698, mag. 2005, doi: 10.1529/biophysj.104.045476.
Minc, N., Burgess, D., and Chang, F. (2011). Influence of cell geometry on division-plane positioning. Cell, 144(3), 414–426.
Khademhosseini, A., Langer, R., Borenstein, J., and Vacanti, J. P. (2006). Microscale technologies for tissue engineering and biology. Proceedings of the National Academy of Sciences, 103(8), 2480–2487.
Schillinger, D., and Ruess, M. (2015). The finite cell method: a review in the context of higher-order structural analysis of CAD and image-based geometric Archives of Computational Methods in Engineering, 22, 391–455.
Murphy, D. B., and Davidson, M. W. (2012). Fundamentals of light microscopy and electronic imaging. John Wiley & Sons.
D. E. Chandler e R. W. Roberson, Bioimaging: Current Concepts in Light and Electron Microscopy. Jones & Bartlett Publishers, 2009.
J. R. Swedlow e K. W. Eliceiri, Open source bioimage informatics for cell biology, Trends in Cell Biology, vol. 19, fasc. 11, pp. 656–660, nov. 2009, doi: 10.1016/j.tcb.2009.08.007.
Mohammed, A., and Abdullah, A. (2018, November). Scanning electron microscopy (SEM): A review. In Proceedings of the 2018 International Conference on Hydraulics and Pneumatics – HERVEX, Bãile Govora, Romania (Vol. 2018, pp. 7–9).
T. R. Jones et al., Scoring diverse cellular morphologies in image-based screens with iterative feedback and machine learning, Proceedings of the National Academy of Sciences, vol. 106, fasc. 6, pp. 1826–1831, feb. 2009, doi: 10.1073/pnas.0808843106.
Schindelin, J., Arganda-Carreras, I., Frise, E., Kaynig, V., Longair, M., Pietzsch, T., … and Cardona, A. (2012). Fiji: an open-source platform for biological-image analysis. Nature methods, 9(7), 676–682.
Carpenter, A. E., Jones, T. R., Lamprecht, M. R., Clarke, C., Kang, I. H., Friman, O., … and Sabatini, D. M. (2006). CellProfiler: image analysis software for identifying and quantifying cell phenotypes. Genome biology, 7, 1–11.
Smith, K., Piccinini, F., Balassa, T., Koos, K., Danka, T., Azizpour, H., and Horvath, P. (2018). Phenotypic image analysis software tools for exploring and understanding big image data from cell-based assays. Cell systems, 6(6), 636–653.
Rejniak, K. A., and Anderson, A. R. (2011). Hybrid models of tumor Wiley Interdisciplinary Reviews: Systems Biology and 3(1), 115–125.
Buttenschön, A., and Edelstein-Keshet, L. (2020). Bridging from single to collective cell migration: A review of models and links to experiments. PLoS computational biology, 16(12), e1008411.
D. P. Tieleman e H. J. C. Berendsen, Molecular dynamics simulations of a fully hydrated dipalmitoylphosphatidylcholine bilayer with different macroscopic boundary conditions and parameters, The Journal of Chemical Physics, vol. 105, fasc. 11, pp. 4871–4880, set. 1996, doi: 10.1063/1.472323.
Deserno, M. (2009). Mesoscopic membrane physics: concepts, simulations, and selected applications. Macromolecular rapid communications, 30(9–10), 752–771.
T. Chumley, R. Feres, L. A. Garcia German, e G. Yablonsky, Revisiting Maxwell-Smoluchowski theory: Low surface roughness in straight channels, Chemical Engineering Science, vol. 284, p. 119477, feb. 2024, doi: 10.1016/j.ces.2023.119477.
J. C. Weaver, Electroporation: A general phenomenon for manipulating cells and tissues, Journal of Cellular Biochemistry, vol. 51, fasc. 4, pp. 426–435, 1993, doi: 10.1002/jcb.2400510407.
Smith, K. C., and Weaver, J. C. (2008). Active mechanisms are needed to describe cell responses to submicrosecond, megavolt-per-meter pulses: cell models for ultrashort pulses. Biophysical journal, 95(4), 1547–1563.
B. Zarghooni, A. Dadgarpour, J. Pourahmadazar, e T. A. Denidni, Supershaped metamaterial unit-cells using the gielis formula, in 2015 IEEE International Symposium on Antennas and Propagation & USNC/URSI National Radio Science Meeting, lug. 2015, pp. 458–459. doi: 10.1109/APS.2015.7304615.
Mescia, L., Chiapperino, M. A., Bia, P., Gielis, J., and Caratelli, D. (2017). Modeling of electroporation induced by pulsed electric fields in irregularly shaped cells. IEEE Transactions on Biomedical Engineering, 65(2), 414–423.
Hufnagel, L., Teleman, A. A., Rouault, H., Cohen, S. M., and Shraiman, B. I. (2007). On the mechanism of wing size determination in fly development. Proceedings of the National Academy of Sciences, 104(10), 3835–3840.
A. Leithner et al., Diversified actin protrusions promote environmental exploration but are dispensable for locomotion of leukocytes, Nat Cell Biol, vol. 18, fasc. 11, pp. 1253–1259, nov. 2016, doi: 10.1038/ncb3426.
Y.-J. Liu et al., Confinement and Low Adhesion Induce Fast Amoeboid Migration of Slow Mesenchymal Cells, Cell, vol. 160, fasc. 4, pp. 659–672, feb. 2015, doi: 10.1016/j.cell.2015.01.007.
Yang, N. J., and Hinner, M. J. (2015). Getting across the cell membrane: an overview for small molecules, peptides, and proteins. Site-Specific Protein Labeling: Methods and Protocols, 29–53.
Hinks, J., Wang, Y., Poh, W. H., Donose, B. C., Thomas, A. W., Wuertz, S., … and Seviour, T. (2014). Modeling cell membrane perturbation by molecules designed for transmembrane electron transfer. Langmuir, 30(9), 2429–2440.
S. J. Marrink, Computational Modeling of Realistic Cell Membranes, Biophysical Journal, vol. 114, fasc. 3, p. 367a, feb. 2018, doi: 10.1016/j.bpj.2017.11.2032.
R. I. Macey, Mathematical Models of Membrane Transport Processes, in Membrane Physiology, T. E. Andreoli, J. F. Hoffman, e D. D. Fanestil, A c. di, Boston, MA: Springer US, 1980, pp. 125–146. doi: 10.1007/978-1-4757-1718-1_7.
Wiczew, D., Szulc, N., and Tarek, M. (2021). Molecular dynamics simulations of the effects of lipid oxidation on the permeability of cell Bioelectrochemistry, 141, 107869.
B. Flaherty, J. P. McGarry, e P. E. McHugh, Mathematical Models of Cell Motility, Cell Biochem Biophys, vol. 49, fasc. 1, pp. 14–28, set. 2007, doi: 10.1007/s12013-007-0045-2.
C. F. Fox, The Structure of Cell Membranes, Scientific American, vol. 226, fasc. 2, pp. 30–39, 1972.
Casares, D., Escribá, P. V., and Rosselló, C. A. (2019). Membrane lipid composition: effect on membrane and organelle structure, function and compartmentalization and therapeutic avenues. International journal of molecular sciences, 20(9), 2167.
Campelo, F., Arnarez, C., Marrink, S. J., and Kozlov, M. M. (2014). Helfrich model of membrane bending: from Gibbs theory of liquid interfaces to membranes as thick anisotropic elastic layers. Advances in colloid and interface science, 208, 25–33.
W. Helfrich, Elastic Properties of Lipid Bilayers: Theory and Possible Experiments, Zeitschrift für Naturforschung C, vol. 28, fasc. 11–12, pp. 693–703, dic. 1973, doi: 10.1515/znc-1973-11-1209.
E. Baesu, R. E. Rudd, J. Belak, e M. McElfresh, Continuum modeling of cell membranes, International Journal of Non-Linear Mechanics, vol. 39, fasc. 3, pp. 369–377, apr. 2004, doi: 10.1016/S0020-7462(02)00193-2.
U. Seifert e R. Lipowsky, Morphology of Vesicles, in Handbook of Biological Physics, vol. 1, Elsevier, 1995, pp. 403–463. doi: 10.1016/S1383-8121(06)80025-4.
Y. Bernard, G. Wheeler, e V.-M. Wheeler, Rigidity and stability of spheres in the Helfrich model, Interfaces and Free Boundaries, vol. 19, fasc. 4, pp. 495–523, gen. 2018, doi: 10.4171/ifb/390.
P. B. Canham, The minimum energy of bending as a possible explanation of the biconcave shape of the human red blood cell, Journal of Theoretical Biology, vol. 26, fasc. 1, pp. 61–81, gen. 1970, doi: 10.1016/S0022-5193(70)80032-7.
B. Seguin e E. Fried, Microphysical derivation of the Canham–Helfrich free-energy density, J. Math. Biol., vol. 68, fasc. 3, pp. 647–665, feb. 2014, doi: 10.1007/s00285-013-0647-9.
F. Campelo e A. Hernández-Machado, Model for Curvature-Driven Pearling Instability in Membranes, Phys. Rev. Lett., vol. 99, fasc. 8, p. 088101, ago. 2007, doi: 10.1103/PhysRevLett.99.088101.
S. B. Dev, D. P. Rabussay, G. Widera, e G. A. Hofmann, Medical applications of electroporation, IEEE Transactions on Plasma Science, vol. 28, fasc. 1, pp. 206–223, feb. 2000, doi: 10.1109/27.842905.
J. L. Young e D. A. Dean, Chapter Three - Electroporation-Mediated Gene Delivery, in Advances in Genetics, vol. 89, L. Huang, D. Liu, e E. Wagner, A c. di, in Nonviral Vectors for Gene Therapy, vol. 89., Academic Press, 2015, pp. 49–88. doi: 10.1016/bs.adgen.2014.10.003.
Vernier, P. T., Sun, Y., and Gundersen, M. A. (2006). Nanoelectropulse-driven membrane perturbation and small molecule permeabilization. BMC cell biology, 7, 1–16.
B. Jakstys, M. Jakutaviciute, D. Uzdavinyte, I. Satkauskiene, e S. Satkauskas, Correlation between the loss of intracellular molecules and cell viability after cell electroporation, Bioelectrochemistry, vol. 135, p. 107550, ott. 2020, doi: 10.1016/j.bioelechem.2020.107550.
B. Franchi e S. Lorenzani, Smoluchowski Equation with Variable Coefficients in Perforated Domains: Homogenization and Applications to Mathematical Models in Medicine, in Harmonic Analysis, Partial Differential Equations and Applications: In Honor of Richard L. Wheeden, S. Chanillo, B. Franchi, G. Lu, C. Perez, e E. T. Sawyer, A c. di, Cham: Springer International Publishing, 2017, pp. 49–67. doi: 10.1007/978-3-319-52742-0_4.
T. Kotnik, L. M. Mir, K. Flisar, M. Puc, e D. Miklavèiè, Cell membrane electropermeabilization by symmetrical bipolar rectangular pulses: Part I. Increased efficiency of permeabilization, Bioelectrochemistry, vol. 54, fasc. 1, pp. 83–90, ago. 2001, doi: 10.1016/S1567-5394(01)00114-1.
S. I. Sukharev, V. A. Klenchin, S. M. Serov, L. V. Chernomordik, e C. YuA, Electroporation and electrophoretic DNA transfer into cells. The effect of DNA interaction with electropores, Biophysical Journal, vol. 63, fasc. 5, pp. 1320–1327, nov. 1992, doi: 10.1016/S0006-3495(92)81709-5.
F. J. Sevilla e M. Sandoval, Smoluchowski diffusion equation for active Brownian swimmers, Phys. Rev. E, vol. 91, fasc. 5, p. 052150, mag. 2015, doi: 10.1103/PhysRevE.91.052150.
Song, Y., Zhang, Y., Shen, T., Bajaj, C. L., McCammon, J. A., and Baker, N. A. (2004). Finite element solution of the steady-state Smoluchowski equation for rate constant calculations. Biophysical Journal, 86(4), 2017–2029.
Bani-Hashemian, M. H. (2011). Accurate and Efficient Solution of the Smoluchowski Equation.
Pan, W., Daily, M., and Baker, N. A. (2015). Numerical calculation of protein-ligand binding rates through solution of the Smoluchowski equation using smoothed particle hydrodynamics. BMC biophysics, 8, 1–12.
Tournier, A. L., Fitzjohn, P. W., and Bates, P. A. (2006). Probability-based model of protein-protein interactions on biological timescales. Algorithms for Molecular Biology, 1, 1–11.
Dukhin, A. S., Ulberg, Z. R., Karamushka, V. I., and Gruzina, T. G. (2010). Peculiarities of live cells’ interaction with micro-and nanoparticles. Advances in colloid and interface science, 159(1), 60–71.
D. J. Beltran-Villegas, R. M. Sehgal, D. Maroudas, D. M. Ford, e M. A. Bevan, A Smoluchowski model of crystallization dynamics of small colloidal clusters, The Journal of Chemical Physics, vol. 135, fasc. 15, p. 154506, ott. 2011, doi: 10.1063/1.3652967.
Egorova, E. M. (1994). The validity of the Smoluchowski equation in electrophoretic studies of lipid membranes. Electrophoresis, 15(1), 1125–1131.
Bhonsle, S. P., Arena, C. B., Sweeney, D. C., and Davalos, R. V. (2015). Mitigation of impedance changes due to electroporation therapy using bursts of high-frequency bipolar pulses. Biomedical engineering online, 14, 1–14.
E. Donath e V. Pastushenko, Electrophoretic study of cell surface properties theory and experimental applicability, Journal of Electroanalytical Chemistry and Interfacial Electrochemistry, vol. 116, pp. 31–40, gen. 1980, doi: 10.1016/S0022-0728(80)80218-X.
Jaramillo, P. (2023). Mathematical and numerical modeling of bilipid membrane electroporation: A phase ordering kinetics approach (Doctoral dissertation, Université de Bordeaux).
R. P. Joshi, A. Mishra, e K. H. Schoenbach, Model Assessment of Cell Membrane Breakdown in Clusters and Tissues Under High-Intensity Electric Pulsing, IEEE Transactions on Plasma Science, vol. 36, fasc. 4, pp. 1680–1688, ago. 2008, doi: 10.1109/TPS.2008.917307.
Kotnik, T., and Miklavèiè, D. (2000). Analytical description of transmembrane voltage induced by electric fields on spheroidal cells. Biophysical Journal, 79(2), 670–679.
G. Pucihar, T. Kotnik, J. Teissié, e D. Miklavèiè, Electropermeabilization of dense cell suspensions, Eur Biophys J, vol. 36, fasc. 3, pp. 173–185, mar. 2007, doi: 10.1007/s00249-006-0115-1.
Y. Chizmadzhev, V. Pastushenko, e V. Markin, The Mechanism of the Excitability of Biological Membranes, Russian Chemical Reviews, vol. 44, p. 993, ott. 2007, doi: 10.1070/RC1975v044n11ABEH002474.
W. Hoiles, V. Krishnamurthy, C. G. Cranfield, e B. Cornell, An Engineered Membrane to Measure Electroporation: Effect of Tethers and Bioelectronic Interface, Biophys J, vol. 107, fasc. 6, pp. 1339–1351, set. 2014, doi: 10.1016/j.bpj.2014.07.056.
S. Movahed e D. Li, Electrokinetic transport through the nanopores in cell membrane during electroporation, Journal of Colloid and Interface Science, vol. 369, fasc. 1, pp. 442–452, mar. 2012, doi: 10.1016/j.jcis.2011.12.039.
Pucihar, G., Kotnik, T., Miklavèiè, D., and Teissié, J. (2008). Kinetics of transmembrane transport of small molecules into electropermeabilized Biophysical journal, 95(6), 2837–2848.
Gajula, K., Gupta, R., and Rai, B. (2020). Multiscale modeling of skin electroporation. Langmuir, 36(24), 6651–6660.
Shamoon, D. (2020). Multiscale and Multiphysics analysis of the deformation of cellular arrangements under an electric-field excitation: application to electroporation (Doctoral dissertation, Université de Bretagne occidentale-Brest).
E. Goldberg, A. Soba, D. Gandía, M. L. Fernández, e C. Suárez, Coupled mathematical modeling of cisplatin electroporation, Bioelectrochemistry, vol. 140, p. 107788, ago. 2021, doi: 10.1016/j.bioelechem.2021.107788.
Merla, C., Denzi, A., Paffi, A., Casciola, M., d’Inzeo, G., Apollonio, F., and Liberti, M. (2012). Novel passive element circuits for microdosimetry of nanosecond pulsed electric fields. IEEE Transactions on Biomedical Engineering, 59(8), 2302–2311.
T. R. Gowrishankar e J. C. Weaver, Electrical behavior and pore accumulation in a multicellular model for conventional and supra-electroporation, Biochemical and Biophysical Research Communications, vol. 349, fasc. 2, pp. 643–653, ott. 2006, doi: 10.1016/j.bbrc.2006.08.097.
Lamberti, P., Tucci, V., Romeo, S., Sannino, A., Scarfì, M. R., and Zeni, O. (2013). nsPEF-induced effects on cell membranes: Use of electrophysical model to optimize experimental design. IEEE Transactions on Dielectrics and Electrical Insulation, 20(4), 1231–1238.