Breast Cancer Dormancy – Lessons Learnt
DOI:
https://doi.org/10.13052/ijts2246-8765.2024.042Keywords:
Breast cancer, dormancy, metastasis, bone microenvironment, bone marrowAbstract
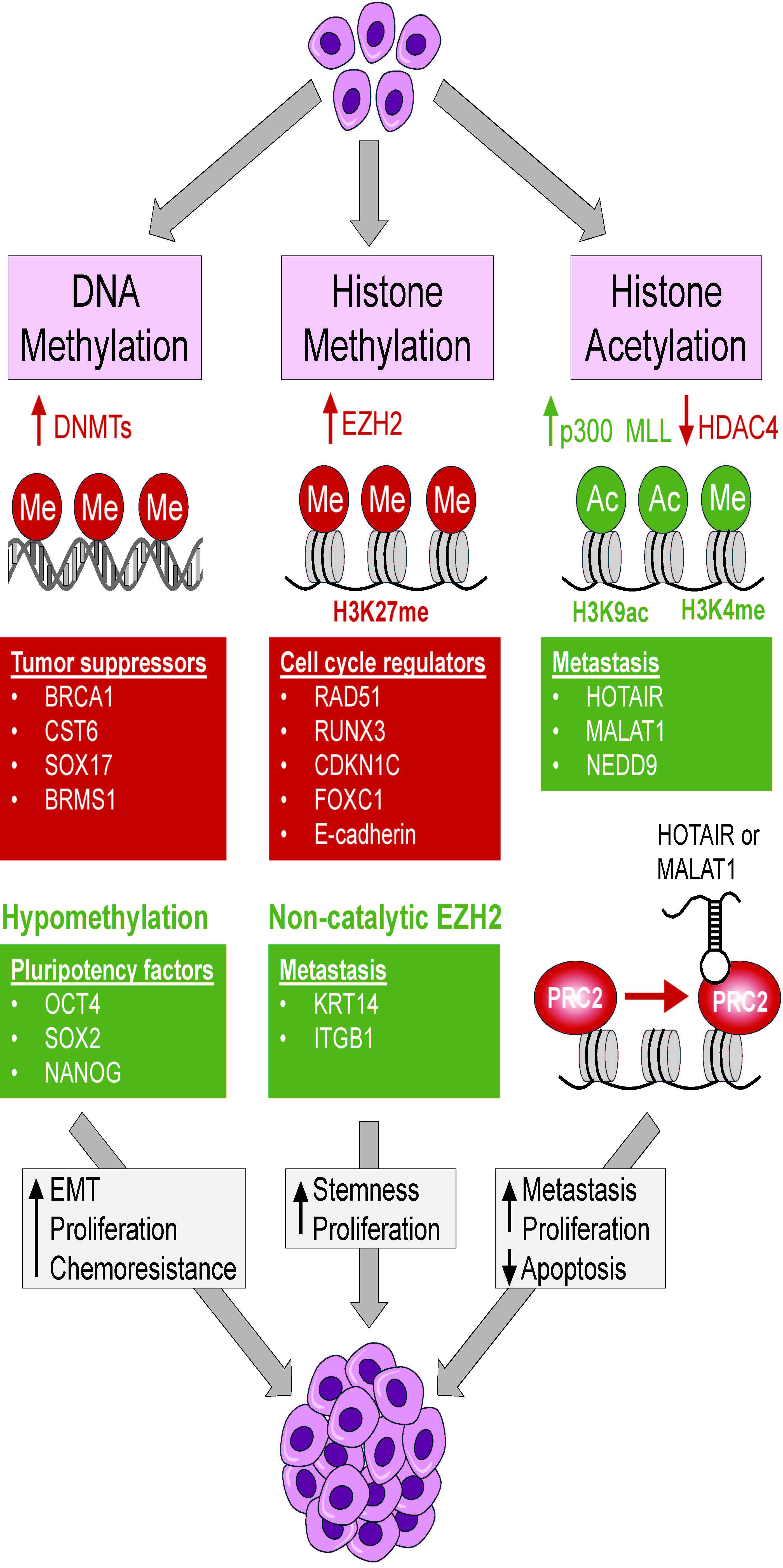
Despite early diagnosis and improved treatments, breast cancer remains a challenging disease. A strategic advantage for breast cancer recurrence is the ability of breast cancer cells to become quiescent and survive for decades in a dormant state within the endosteal region of the bone marrow. Breast cancer dormancy is triggered by exosome vesicles secreted by mesenchymal stem cells residing in the bone marrow. By mechanisms yet to be determined, dormant breast cancer cells are awakened leading to resurgence and metastasis. Experimental evidence supports the notion that dormant breast cancer cells are cancer stem cells recognized as tumor initiating and propagating cells with chemoresistant and metastatic properties. These cells represent less than 2% of the total tumor mass, which impose a significant barrier for their therapeutic targeting. This review focuses on cellular and molecular properties of breast cancer dormancy including tumor microenvironment, epigenetic regulation, cell signaling and metabolic reprogramming.
Downloads
References
Barzaman, K., et al., Breast cancer: Biology, biomarkers, and treatments. Int Immunopharmacol, 2020. 84: p. 106535.
Zagami, P. and L.A. Carey, Triple negative breast cancer: Pitfalls and progress. NPJ Breast Cancer, 2022. 8(1): p. 95.
Zhang, X., K. Powell, and L. Li, Breast Cancer Stem Cells: Biomarkers, Identification and Isolation Methods, Regulating Mechanisms, Cellular Origin, and Beyond. Cancers (Basel), 2020. 12(12).
Talukdar, S.B., P.; Emdad, L.; Das, S.; Sarkar, D.; Fisher, P.B., Dormancy and cancer stem cells: An enigma for cancer therapeutic targeting. Adv. Cancer Res., 2019. 141: p. 43–84.
Ferrer, A.I., Trinidad, J.R., Sandiford, O. et al., Epigenetic dynamics in cancer stem cell dormancy. Cancer Metastasis Rev, 2020. 39: p. 721–738.
Sowder, M.E., and Rachelle W Johnson, Bone as a Preferential Site for Metastasis. JBMR, 2019. 3(3).
Eltoukhy, H.S., et al., Secretome within the bone marrow microenvironment: A basis for mesenchymal stem cell treatment and role in cancer dormancy. Biochimie, 2018. 155: p. 92–103.
Walker, N.D., et al., The bone marrow niche in support of breast cancer dormancy. Cancer Lett, 2016. 380(1): p. 263–71.
Willis, R.A., The Spread of Tumours in the Human Body. 1934, London: J. and A. Churchill.
Hadfield, G., The dormant cancer cell. Br Med J, 1954. 2(4888): p. 607–10.
Dalla, E., et al., Dormancy in Breast Cancer. Cold Spring Harb Perspect Med, 2023. 13(11).
Tachtsidis, A., et al., Minimal residual disease in breast cancer: an overview of circulating and disseminated tumour cells. Clin Exp Metastasis, 2016. 33(6): p. 521–50.
Hartkopf, A.D., et al., Disseminated tumour cells from the bone marrow of early breast cancer patients: Results from an international pooled analysis. Eur J Cancer, 2021. 154: p. 128–137.
Bidard, F.C., et al., Disseminated tumor cells of breast cancer patients: a strong prognostic factor for distant and local relapse. Clin Cancer Res, 2008. 14(11): p. 3306–11.
Hartkopf, A.D., et al., Disseminated tumor cells from the bone marrow of patients with nonmetastatic primary breast cancer are predictive of locoregional relapse. Ann Oncol, 2015. 26(6): p. 1155–1160.
Wiedswang, G., et al., Detection of isolated tumor cells in bone marrow is an independent prognostic factor in breast cancer. J Clin Oncol, 2003. 21(18): p. 3469–78.
Aguirre-Ghiso, J.A., Models, mechanisms and clinical evidence for cancer dormancy. Nat Rev Cancer, 2007. 7(11): p. 834–46.
Weidenfeld, K. and D. Barkan, EMT and Stemness in Tumor Dormancy and Outgrowth: Are They Intertwined Processes? Front Oncol, 2018. 8: p. 381.
Reddy, B.Y., et al., The Microenvironmental Effect in the Progression, Metastasis, and Dormancy of Breast Cancer: A Model System within Bone Marrow. Int J Breast Cancer, 2012. 2012: p. 721659.
Casson, J., et al., Mesenchymal stem cell-derived extracellular vesicles may promote breast cancer cell dormancy. J Tissue Eng, 2018. 9: p. 2041731418810093.
Mani, S.A., et al., The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell, 2008. 133(4): p. 704–15.
Plaks, V., N. Kong, and Z. Werb, The cancer stem cell niche: how essential is the niche in regulating stemness of tumor cells? Cell Stem Cell, 2015. 16(3): p. 225–38.
Brabletz, S., et al., Dynamic EMT: a multi-tool for tumor progression. EMBO J, 2021. 40(18): p. e108647.
Aouad, P., et al., Epithelial-mesenchymal plasticity determines estrogen receptor positive breast cancer dormancy and epithelial reconversion drives recurrence. Nat Commun, 2022. 13(1): p. 4975.
Li, Y., et al., The role of cancer-associated fibroblasts in breast cancer metastasis. Front Oncol, 2023. 13: p. 1194835.
Butti, R., et al., Tumor-derived osteopontin drives the resident fibroblast to myofibroblast differentiation through Twist1 to promote breast cancer progression. Oncogene, 2021. 40(11): p. 2002–2017.
Sjoberg, E., et al., A Novel ACKR2-Dependent Role of Fibroblast-Derived CXCL14 in Epithelial-to-Mesenchymal Transition and Metastasis of Breast Cancer. Clin Cancer Res, 2019. 25(12): p. 3702–3717.
Yu, Y., et al., Cancer-associated fibroblasts induce epithelial-mesenchymal transition of breast cancer cells through paracrine TGF-beta signalling. Br J Cancer, 2014. 110(3): p. 724–32.
Mehta, A.K., et al., Macrophage Biology and Mechanisms of Immune Suppression in Breast Cancer. Front Immunol, 2021. 12: p. 643771.
Jacob, A. and R. Prekeris, The regulation of MMP targeting to invadopodia during cancer metastasis. Front Cell Dev Biol, 2015. 3: p. 4.
Tian, K., et al., MMP-9 secreted by M2-type macrophages promotes Wilms’ tumour metastasis through the PI3K/AKT pathway. Mol Biol Rep, 2022. 49(5): p. 3469–3480.
Wang, N., et al., CXCL1 derived from tumor-associated macrophages promotes breast cancer metastasis via activating NF-kappaB/SOX4 signaling. Cell Death Dis, 2018. 9(9): p. 880.
Pulido, C., et al., Bone metastasis risk factors in breast cancer. Ecancermedicalscience, 2017. 11: p. 715.
Paget, S., The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev, 1989. 8(2): p. 98–101.
Ema, H. and T. Suda, Two anatomically distinct niches regulate stem cell activity. Blood, 2012. 120(11): p. 2174–81.
Patel, S.A., et al., Delineation of breast cancer cell hierarchy identifies the subset responsible for dormancy. Sci Rep, 2012. 2: p. 906.
Sinha, G., et al., Gap Junctions and Breast Cancer Dormancy. Trends Cancer, 2020. 6(4): p. 348–357.
Aasen, T., et al., Gap junctions and cancer: communicating for 50 years. Nat Rev Cancer, 2016. 16(12): p. 775–788.
Oshima, A., Structure and closure of connexin gap junction channels. FEBS Lett, 2014. 588(8): p. 1230–7.
Lim, P.K., et al., Gap junction-mediated import of microRNA from bone marrow stromal cells can elicit cell cycle quiescence in breast cancer cells. Cancer Res, 2011. 71(5): p. 1550–60.
Park, J.M., et al., Exogenous CXCL12 activates protein kinase C to phosphorylate connexin 43 for gap junctional intercellular communication among confluent breast cancer cells. Cancer Lett, 2013. 331(1): p. 84–91.
Moharita, A.L., et al., SDF-1alpha regulation in breast cancer cells contacting bone marrow stroma is critical for normal hematopoiesis. Blood, 2006. 108(10): p. 3245–52.
Ferrer, A.I., et al., Epigenetic dynamics in cancer stem cell dormancy. Cancer Metastasis Rev, 2020. 39(3): p. 721–738.
Ono, M., et al., Exosomes from bone marrow mesenchymal stem cells contain a microRNA that promotes dormancy in metastatic breast cancer cells. Sci Signal, 2014. 7(332): p. ra63.
Bliss, S.A., et al., Mesenchymal Stem Cell-Derived Exosomes Stimulate Cycling Quiescence and Early Breast Cancer Dormancy in Bone Marrow. Cancer Res, 2016. 76(19): p. 5832–5844.
Sandiford, O.A., et al., Mesenchymal Stem Cell-Secreted Extracellular Vesicles Instruct Stepwise Dedifferentiation of Breast Cancer Cells into Dormancy at the Bone Marrow Perivascular Region. Cancer Res, 2021. 81(6): p. 1567–1582.
Nobre, A.R., et al., Bone marrow NG2(+)/Nestin(+) mesenchymal stem cells drive DTC dormancy via TGFbeta2. Nat Cancer, 2021. 2(3): p. 327–339.
Walker, N.D., et al., Exosomes from differentially activated macrophages influence dormancy or resurgence of breast cancer cells within bone marrow stroma. Cell Death Dis, 2019. 10(2): p. 59.
Montesinos, J.J., et al., In vitro evidence of the presence of mesenchymal stromal cells in cervical cancer and their role in protecting cancer cells from cytotoxic T cell activity. Stem Cells Dev, 2013. 22(18): p. 2508–19.
Hsu, W.T., et al., Prostaglandin E2 potentiates mesenchymal stem cell-induced IL-10+IFN-gamma+CD4+ regulatory T cells to control transplant arteriosclerosis. J Immunol, 2013. 190(5): p. 2372–80.
Bernardo, M.E. and W.E. Fibbe, Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell, 2013. 13(4): p. 392–402.
Waddington, C.H., The epigenotype. 1942. Int J Epidemiol, 2012. 41(1): p. 10–3.
Crea, F., et al., The epigenetic/noncoding origin of tumor dormancy. Trends Mol Med, 2015. 21(4): p. 206–11.
Moore, L.D., T. Le, and G. Fan, DNA methylation and its basic function. Neuropsychopharmacology, 2013. 38(1): p. 23–38.
Lyko, F., The DNA methyltransferase family: a versatile toolkit for epigenetic regulation. Nat Rev Genet, 2018. 19(2): p. 81–92.
Lio, C.J., et al., TET methylcytosine oxidases: new insights from a decade of research. J Biosci, 2020. 45.
Suva, M.L., N. Riggi, and B.E. Bernstein, Epigenetic reprogramming in cancer. Science, 2013. 339(6127): p. 1567–70.
Hurtado, A., et al., FOXA1 is a key determinant of estrogen receptor function and endocrine response. Nat Genet, 2011. 43(1): p. 27–33.
Mirza, S., et al., Promoter hypermethylation of TMS1, BRCA1, ERalpha and PRB in serum and tumor DNA of invasive ductal breast carcinoma patients. Life Sci, 2007. 81(4): p. 280–7.
Shargh, S.A., et al., Downregulation of E-cadherin expression in breast cancer by promoter hypermethylation and its relation with progression and prognosis of tumor. Med Oncol, 2014. 31(11): p. 250.
Theodorou, V., et al., GATA3 acts upstream of FOXA1 in mediating ESR1 binding by shaping enhancer accessibility. Genome Res, 2013. 23(1): p. 12–22.
Zhu, X., et al., Hypermethylation of BRCA1 gene: implication for prognostic biomarker and therapeutic target in sporadic primary triple-negative breast cancer. Breast Cancer Res Treat, 2015. 150(3): p. 479–86.
Esteller, M., et al., Promoter hypermethylation and BRCA1 inactivation in sporadic breast and ovarian tumors. J Natl Cancer Inst, 2000. 92(7): p. 564–9.
Liang, F., et al., Liquiritigenin decreases tumorigenesis by inhibiting DNMT activity and increasing BRCA1 transcriptional activity in triple-negative breast cancer. Exp Biol Med (Maywood), 2021. 246(4): p. 459–466.
Zhang, J., et al., Correlation between promoter methylation of the LDH-C4 gene and DNMT expression in breast cancer and their prognostic significance. Oncol Lett, 2022. 23(1): p. 35.
Fu, Y., et al., The DNMT1-PAS1-PH20 axis drives breast cancer growth and metastasis. Signal Transduct Target Ther, 2022. 7(1): p. 81.
Liu, X., et al., The EZH2- H3K27me3-DNMT1 complex orchestrates epigenetic silencing of the wwc1 gene, a Hippo/YAP pathway upstream effector, in breast cancer epithelial cells. Cell Signal, 2018. 51: p. 243–256.
Chimonidou, M., et al., DNA methylation of tumor suppressor and metastasis suppressor genes in circulating tumor cells. Clin Chem, 2011. 57(8): p. 1169–77.
Gkountela, S., et al., Circulating Tumor Cell Clustering Shapes DNA Methylation to Enable Metastasis Seeding. Cell, 2019. 176(1–2): p. 98–112 e14.
Shyam A. Patel, S.H.R., Margarette Bryan, Lillian F. Pliner, Gabriela Dontu, Prem S. Patel, Sohrab Amiri, Sharon R. Pine, and Pranela Rameshwar, Delineation of breast cancer cell hierachy identifies the subset responsible for dormancy. 2012.
Vasefifar, P., et al., Nanog, as a key cancer stem cell marker in tumor progression. Gene, 2022. 827: p. 146448.
Kim, K.H. and C.W. Roberts, Targeting EZH2 in cancer. Nat Med, 2016. 22(2): p. 128–34.
Yoo, K.H. and L. Hennighausen, EZH2 methyltransferase and H3K27 methylation in breast cancer. Int J Biol Sci, 2012. 8(1): p. 59–65.
Verma, A., et al., EZH2-H3K27me3 mediated KRT14 upregulation promotes TNBC peritoneal metastasis. Nat Commun, 2022. 13(1): p. 7344.
Zhang, L., et al., EZH2 engages TGFbeta signaling to promote breast cancer bone metastasis via integrin beta1-FAK activation. Nat Commun, 2022. 13(1): p. 2543.
Guo, P., et al., The Histone Acetylation Modifications of Breast Cancer and their Therapeutic Implications. Pathol Oncol Res, 2018. 24(4): p. 807–813.
Bhan, A., et al., Antisense transcript long noncoding RNA (lncRNA) HOTAIR is transcriptionally induced by estradiol. J Mol Biol, 2013. 425(19): p. 3707–22.
Hu, Z., et al., Histone deacetylase inhibitors promote breast cancer metastasis by elevating NEDD9 expression. Signal Transduct Target Ther, 2023. 8(1): p. 11.
Leslie, P.L., et al., Histone deacetylase 11 inhibition promotes breast cancer metastasis from lymph nodes. Nat Commun, 2019. 10(1): p. 4192.
Kumar, S., et al., Non-Coding RNAs as Mediators of Epigenetic Changes in Malignancies. Cancers (Basel), 2020. 12(12).
Panjarian, S. and J.J. Issa, The Roles of DNA Demethylases in Triple-Negative Breast Cancer. Pharmaceuticals (Basel), 2021. 14(7).
Han, M., et al., MiR-21 regulates epithelial-mesenchymal transition phenotype and hypoxia-inducible factor-1alpha expression in third-sphere forming breast cancer stem cell-like cells. Cancer Sci, 2012. 103(6): p. 1058–64.
Mei, M., et al., Downregulation of miR-21 enhances chemotherapeutic effect of taxol in breast carcinoma cells. Technol Cancer Res Treat, 2010. 9(1): p. 77–86.
Gupta, R.A., et al., Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature, 2010. 464(7291): p. 1071–6.
Jadaliha, M., et al., Functional and prognostic significance of long non-coding RNA MALAT1 as a metastasis driver in ER negative lymph node negative breast cancer. Oncotarget, 2016. 7(26): p. 40418–40436.
Sanchez Calle, A., et al., Long non-coding NR2F1-AS1 is associated with tumor recurrence in estrogen receptor-positive breast cancers. Mol Oncol, 2020. 14(9): p. 2271–2287.
Fukuoka, M., et al., The ELEANOR noncoding RNA expression contributes to cancer dormancy and predicts late recurrence of estrogen receptor-positive breast cancer. Cancer Sci, 2022. 113(7): p. 2336–2351.
Hanahan, D. and R.A. Weinberg, The hallmarks of cancer. Cell, 2000. 100(1): p. 57–70.
Pranzini, E., G. Raugei, and M.L. Taddei, Metabolic Features of Tumor Dormancy: Possible Therapeutic Strategies. Cancers (Basel), 2022. 14(3).
Ferrer, A., et al., Hypoxia-mediated changes in bone marrow microenvironment in breast cancer dormancy. Cancer Lett, 2020. 488: p. 9–17.
Martins Pinto, M., et al., The Warburg effect and mitochondrial oxidative phosphorylation: Friends or foes? Biochim Biophys Acta Bioenerg, 2023. 1864(1): p. 148931.
Koppenol, W.H., P.L. Bounds, and C.V. Dang, Otto Warburg’s contributions to current concepts of cancer metabolism. Nat Rev Cancer, 2011. 11(5): p. 325–37.
Ruidas, B., Mitochondrial lipid metabolism in metastatic breast cancer. 2024.
Schomel, N., et al., UGCG influences glutamine metabolism of breast cancer cells. Sci Rep, 2019. 9(1): p. 15665.
Schomel, N., et al., UGCG overexpression leads to increased glycolysis and increased oxidative phosphorylation of breast cancer cells. Sci Rep, 2020. 10(1): p. 8182.
Zheng, J., Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation (Review). Oncol Lett, 2012. 4(6): p. 1151–1157.
Buschhaus, J.M., et al., Targeting disseminated estrogen-receptor-positive breast cancer cells in bone marrow. Oncogene, 2020. 39(34): p. 5649–5662.
Martini, M., et al., PI3K/AKT signaling pathway and cancer: an updated review. Ann Med, 2014. 46(6): p. 372–83.
Sanchez-Alvarez, R., et al., Mitochondrial Fission Factor (MFF) Inhibits Mitochondrial Metabolism and Reduces Breast Cancer Stem Cell (CSC) Activity. Front Oncol, 2020. 10: p. 1776.
Tau, S. and T.W. Miller, The role of cancer cell bioenergetics in dormancy and drug resistance. Cancer Metastasis Rev, 2023. 42(1): p. 87–98.
Munoz, D., et al., Effects of screening and systemic adjuvant therapy on ER-specific US breast cancer mortality. J Natl Cancer Inst, 2014. 106(11).
Wang, W., et al., Hypoxia-inducible factor 1alpha in breast cancer prognosis. Clin Chim Acta, 2014. 428: p. 32–7.
Paredes, F., H.C. Williams, and A. San Martin, Metabolic adaptation in hypoxia and cancer. Cancer Lett, 2021. 502: p. 133–142.
Alejandra Ferrer, J.R.T., Oleta Sandiford,, Jean-Pierre Etchegaray, and Pranela Rameshwar, Epigenetic dynamics in cancer stem cell dormancy. 2020.
Davis, J.E., Jr., et al., Tumor Dormancy and Slow-Cycling Cancer Cells. Adv Exp Med Biol, 2019. 1164: p. 199–206.
Chen, A., et al., Intermittent hypoxia induces a metastatic phenotype in breast cancer. Oncogene, 2018. 37(31): p. 4214–4225.
Thienpont, B., et al., Tumour hypoxia causes DNA hypermethylation by reducing TET activity. Nature, 2016. 537(7618): p. 63–68.
Pohl, S.G., et al., Wnt signaling in triple-negative breast cancer. Oncogenesis, 2017. 6(4): p. e310.
Harper, K.L., et al., Mechanism of early dissemination and metastasis in Her2(+) mammary cancer. Nature, 2016. 540(7634): p. 588–592.
Yin, P., et al., Wnt signaling in human and mouse breast cancer: Focusing on Wnt ligands, receptors and antagonists. Cancer Sci, 2018. 109(11): p. 3368–3375.
Malladi, S., et al., Metastatic Latency and Immune Evasion through Autocrine Inhibition of WNT. Cell, 2016. 165(1): p. 45–60.
Bushnell, G.G., et al., Breast cancer dormancy: need for clinically relevant models to address current gaps in knowledge. NPJ Breast Cancer, 2021. 7(1): p. 66.
Fiorillo, M., et al., High ATP Production Fuels Cancer Drug Resistance and Metastasis: Implications for Mitochondrial ATP Depletion Therapy. Front Oncol, 2021. 11: p. 740720.
Nielsen, T.O., et al., Assessment of Ki67 in Breast Cancer: Updated Recommendations From the International Ki67 in Breast Cancer Working Group. J Natl Cancer Inst, 2021. 113(7): p. 808–819.
Bartlome, S. and C.C. Berry, Recent insights into the effects of metabolism on breast cancer cell dormancy. Br J Cancer, 2022. 127(8): p. 1385–1393.