Specific Binding of Cancer-derived Exosomes to Synthetic Carbon Beads – Implication for Adjuvant and Neoadjuvant Cancer Treatment
DOI:
https://doi.org/10.13052/ijts2246-8765.2025.004Keywords:
Carbon, cancer, stem cell, exosomes, glioblastoma, ovarian cancerAbstract
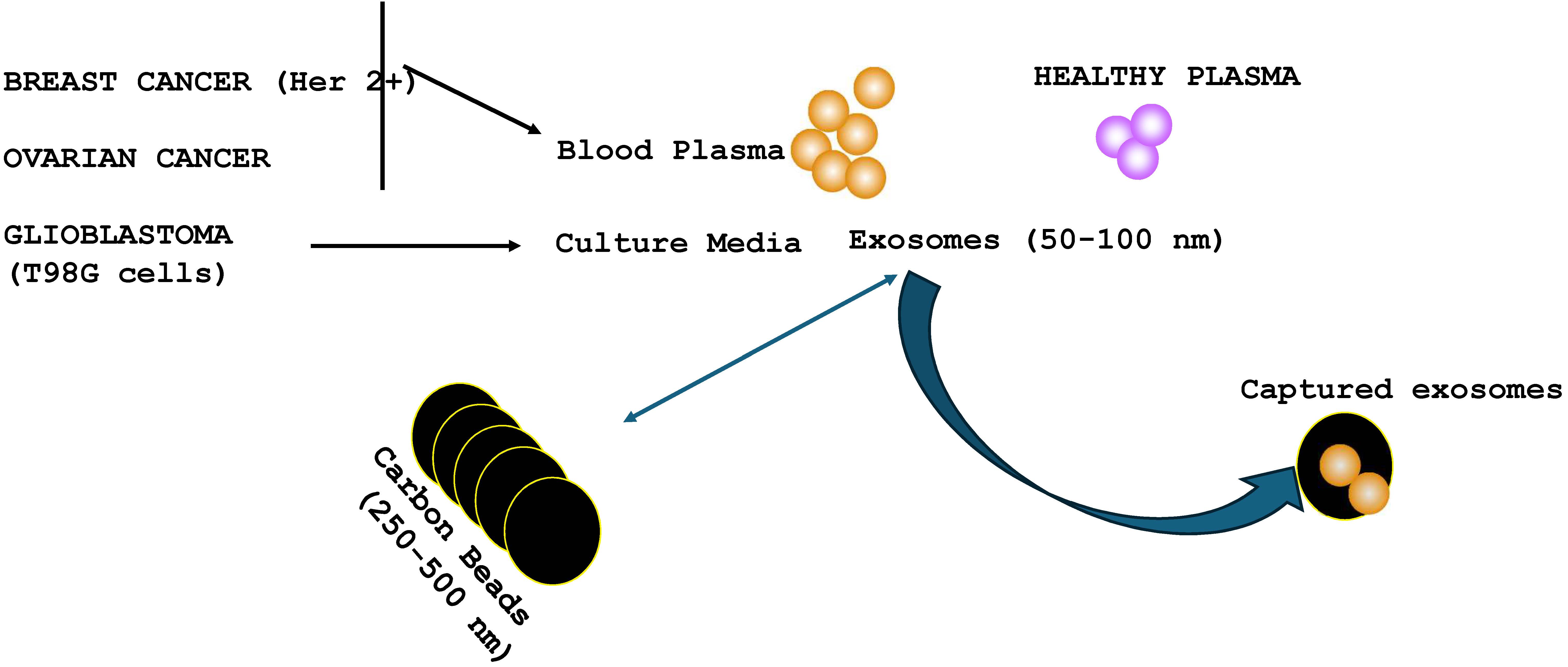
Extracellular vesicles (exosomes) can mediate intercellular communication. In cancer patients, exosomes are central to intercellular communication between cancer cells and non-malignant cells within the tissue niche, and at distant regions. This type of communication influences the biology of the cancer and treatment response. This study tested the hypothesis that carbon beads, 150–500 μm in size with pore size about 27 nm, can capture exosomes from the plasma of cancer patients, and from a glioblastoma cell line (T98G). The challenge to addressing the hypothesis is the relatively larger size of exosomes, 50–120 nm, to be captured within the carbon pore. This study tested plasma from a patient with ovarian cancer and two breast cancer patients, HER2+ and triple negative for hormone receptor (TNBC), and released exosomes from T98G cells. This study showed exosomes from ovarian plasma binding to a specific synthetic carbon, C529. Similar binding did not occur with plasma from age-matched healthy controls. GBM exosomes also bound to C529 but the repertoire of carbon was broader. TNBC exosomes showed low affinity for the identified carbon. However, exosomes from HER2+ breast cancer were able to bind to the carbon beads. In total, this study showed binding of exosomes from cancer plasma to C529 carbon. The results of these findings are discussed in the context of potential treatment, and they provide future direction for studies to examine carbon beads as adjuvant or neoadjuvant treatment for cancer.
Downloads
References
Bang, C., and T. Thum. 2012. Exosomes: new players in cell–cell communication. The Intl J Viochem Cell Biol 44: 2060–2064.
Sandiford, O. A., R. J. Donnelly, M. H. El-Far, L. M. Burgmeyer, G. Sinha, S. H. Pamarthi, L. S. Sherman, A. I. Ferrer, D. E. DeVore, S. A. Patel, Y. Naaldijk, S. Alonso, P. Barak, M. Bryan, N. M. Ponzio, R. Narayanan, J. P. Etchegaray, R. Kumar, and P. Rameshwar. 2021. Mesenchymal Stem Cell-Secreted Extracellular Vesicles Instruct Stepwise Dedifferentiation of Breast Cancer Cells into Dormancy at the Bone Marrow Perivascular Region. Cancer Res 81: 1567–1582.
Walker, N. D., M. Elias, K. Guiro, R. Bhatia, S. J. Greco, M. Bryan, M. Gergues, O. A. Sandiford, N. M. Ponzio, S. J. Leibovich, and P. Rameshwar. 2019. Exosomes from differentially activated macrophages influence dormancy or resurgence of breast cancer cells within bone marrow stroma. Cell Death Dis 10: 59.
Bliss, S. A., G. Sinha, O. A. Sandiford, L. M. Williams, D. J. Engelberth, K. Guiro, L. L. Isenalumhe, S. J. Greco, S. Ayer, M. Bryan, R. Kumar, N. M. Ponzio, and P. Rameshwar. 2016. Mesenchymal Stem Cell-Derived Exosomes Stimulate Cycling Quiescence and Early Breast Cancer Dormancy in Bone Marrow. Cancer Res 76: 5832–5844.
Greco, S. J., S. Ayer, K. Guiro, G. Sinha, R. J. Donnelly, M. H. El-Far, L. S. Sherman, Y. Kenfack, S. H. Pamarthi, M. Gergues, O. A. Sandiford, M. J. Schonning, J. P. Etchegaray, and P. Rameshwar. 2021. Restoration of aged hematopoietic cells by their young counterparts through instructive microvesicles release. Aging (Albany NY) 13: 23981–24016.
Buschow, S. I., B. W. Van Balkom, M. Aalberts, A. J. Heck, M. Wauben, and W. Stoorvogel. 2010. MHC class II-associated proteins in B-cell exosomes and potential functional implications for exosome biogenesis. Immunol cell Biol 88: 851–856.
Sun, B., P. Dalvi, L. Abadjian, N. Tang, and L. Pulliam. 2017. Blood neuron-derived exosomes as biomarkers of cognitive impairment in HIV. Aids 31: F9–F17.
Paskeh, M. D. A., M. Entezari, S. Mirzaei, A. Zabolian, H. Saleki, M. J. Naghdi, S. Sabet, M. A. Khoshbakht, M. Hashemi, and K. Hushmandi. 2022. Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. J Hematol Oncol 15: 83.
Kalluri, R. 2016. The biology and function of exosomes in cancer. J Clin Invest 126: 1208–1215.
Potian, J. A., H. Aviv, N. M. Ponzio, J. S. Harrison, and P. Rameshwar. 2003. Veto-like activity of mesenchymal stem cells: functional discrimination between cellular responses to alloantigens and recall antigens. J Immunol 171: 3426–3434.
Munoz, J. L., S. A. Bliss, S. J. Greco, S. H. Ramkissoon, K. L. Ligon, and P. Rameshwar. 2013. Delivery of Functional Anti-miR-9 by Mesenchymal Stem Cell-derived Exosomes to Glioblastoma Multiforme Cells Conferred Chemosensitivity. Mol Ther Nucleic Acids 2: e126.
Xu, X., F. Yin, M. Guo, G. Gan, G. Lin, C. Wen, J. Wang, P. Song, J. Wang, Z. Q. Qi, and C. Q. Zhong. 2023. Quantitative proteomic analysis of exosomes from umbilical cord mesenchymal stem cells and rat bone marrow stem cells. Proteomics 23: e2200204.
Liu, S.-L., P. Sun, Y. Li, S.-S. Liu, and Y. Lu. 2019. Exosomes as critical mediators of cell-to-cell communication in cancer pathogenesis and their potential clinical application. Transl Cancer Res 8: 298.
Anker, P., H. Mulcahy, X. Qi Chen, and M. Stroun. 1999. Detection of circulating tumour DNA in the blood (plasma/serum) of cancer patients. Cancer Met Rev 18: 65–73.
Thakur, B. K., H. Zhang, A. Becker, I. Matei, Y. Huang, B. Costa-Silva, Y. Zheng, A. Hoshino, H. Brazier, and J. Xiang. 2014. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res 24: 766–769.
Gemel, J., J. Kilkus, G. Dawson, and E. C. Beyer. 2019. Connecting Exosomes and Connexins. Cancers 11: 476.
Soares, A. R., T. Martins-Marques, T. Ribeiro-Rodrigues, J. V. Ferreira, S. Catarino, M. J. Pinho, M. Zuzarte, S. Isabel Anjo, B. Manadas, J. P.G. Sluijter, P. Pereira, and H. Girao. 2015. Gap junctional protein Cx43 is involved in the communication between extracellular vesicles and mammalian cells. Sci Reports 5: 13243.
Norris, R. P. 2021. Transfer of mitochondria and endosomes between cells by gap junction internalization. Traffic 22: 174–179.
Record, M., K. Carayon, M. Poirot, and S. Silvente-Poirot. 2014. Exosomes as new vesicular lipid transporters involved in cell–cell communication and various pathophysiologies. Biochimica et Biophysica Acta (BBA)-Mol Cell Biol Lipids 1841: 108–120.
Zhang, P., Q. Xia, L. Liu, S. Li, and L. Dong. 2020. Current opinion on molecular characterization for GBM classification in guiding clinical diagnosis, prognosis, and therapy. Frontiers Mol Biosci 7: 562798.
Au, K. K., J. A. Josahkian, J.-A. Francis, J. A. Squire, and M. Koti. 2015. Current state of biomarkers in ovarian cancer prognosis. Future Oncol 11: 3187–3195.